Abstract
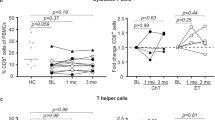
Natural killer (NK) cell activity, the autologous mixed lymphocyte reaction (AMLR) and proportions of T cell subpopulations (CD3+/CD4+ and CD3+/CD8+) and NK cells (CD16+) were studied in 21 patients with bilateral primary breast cancer (BBC), 10 patients with single-breast cancer (SBC) and 20 healthy controls. All patients studied had no evidence of disease and had been off radiotherapy and/or chemotherapy for at least 1 year. Ten patients with BBC were also treated with tamoxifen. Patients with SBC had NK cell activity, AMLR responses and T cell subpopulations that were comparable to those of normal controls. In patients with BBC, a significant (P<0.01) increase in NK activity compared to that in normal controls (42±13% versus 21±10%, effector-to-target cell ratio, 25∶1) and a significant (P<0.05) decrease in CD4+ T cell proportions (30±15% versus 49±13%) and absolute numbers (472±82/mm3 versus 953±131/mm3) were found. However, the proliferative response of BBC patients' T lymphocytes in AMLR was in the range of the normal controls. Lymphocytes derived from 10 BBC patients treated with tamoxifen exhibited NK cell activity that was comparable to that of normal controls and patients with SBC, and was significantly (P<0.01) reduced compared to the pretreatment period. BBC patients who received tamoxifen also show a reduction in the proportion of CD4+ T cells and in AMLR proliferative responses, which decreased compared to levels in normal controls. Taken together, these results indicate that long-term tamoxifen treatment modulates immune responses in BBC patients.
Similar content being viewed by others
References
Berry J, Green BJ, Matheson DS (1987) Modulation of natural killer cell activity by tamoxifen in stage I post-menopause breast cancer. Eur J Cancer Clin Oncol 23: 517
Bonilla F, Alvares-Mon M, Merino P, Giron JA, Mendez SR, Espana P, Durantez A (1990) Natural killer activity in patients with breast cancer. Eur J Gynaecol Oncol 11: 103
Brenner BG, Friedman G, Margolese RG (1985) The relationship of clinical status and therapeutic modality to NK cell activity in human breast cancer. Cancer 56: 1543
Cannon NE, Bonnard G, Ojen W, Herberman R (1977) Relationship of human natural lymphocyte mediated cytotoxicity of breast cancer derived target cells. Int J Cancer 19: 487
Cunningham-Rundles S, Filippa DA, Braun DW, Antonelli P, Ashikori F (1981) Natural cytotoxicity of peripheral blood lymphocytes and regional lymph node cells in breast cancer in women. JNCI 67: 585
Editorial (1988) Second primary breast cancer. Br Med J 296: 1755
Ermin O, Ashby J, Stephens J (1978) Human natural cytotoxicity in the blood and lymph organs of healthy donors and patients with malignant diseases. Int J Cancer 21: 35
Friedman-Birnbaum R, Weltfriend S, Pollack S (1991) Classic Kaposi sarcoma: T lymphocyte subsets and NK cell activity. J Am Acad Dermatol 24: 937
Harvey EB, Brinton LA (1985) Second cancer following cancer of the breast in Connecticut, 1935–82. NCI Monogr 68: 99
Henderson JC (1987) Adjuvant systemic therapy for early breast cancer. Curr Probl Cancer 11: 125
Joensun H, Toivanen A, Nordman E (1986) Effect of tamoxifen on immune functions. Cancer Treat Rep 70: 381
Mackay IR, Googjear MDE, Riglar C, Penschow J (1983) Effect of natural killer antibody dependent cellular cytotoxicity of adjuvant cytostatic chemotherapy including melphalan in breast cancer. Cancer Immunnol Immunother 16: 98
Myers MJ, Butler LD, Petersen BH (1986) Estradiol-induced alteration in the immune system. II. Suppression of cellular immunity in the rat is not the result of direct estrogenic action. Immunopharmacology 11: 47
Nolvadex Adjuvant Trial Organization (1987) Controlled trial of tamoxifen as single adjuvant agent in the management of early breast cancer: analysis of six years. Lancet I: 836
Robinson E, Mekori T, Segal R, Nasarllah S (1993) The T helper and suppressor lymphocyte subset in patients with multiple primary cancers. Oncology (in press)
Romain PL, Lipsky PE (1983) Immunoglobulin secretion during the autologous mixed lymphocyte reaction in man. J Immunol 130: 1146
Rotstein S, Blomgren H, Petrini B, Wasserman J, Von Steding LV (1988) Influence of adjuvant tamoxifen on blood lymphocytes. Breast Cancer Res Treat 12: 75
Rouse BT, Horohov DW (1986) Immunosuppression in viral infections. Rev Infect Dis 8: 850
Sheard CR, Reiley F, Tee DEH, Vergani D, Lowe D, Baum M, Cameron AEP (1986) The effect of adjuvant cyclophosphamide and tamoxifen on the number of lymphocytes bearing T cells or NK cells. Br J Cancer 54: 705
Tartter PI, Steinberg B, Barron D, Martinelli G (1987) The prognostic significance of natural killer cytotoxicity in patients with colorectal cancer. Arch Surg 122: 1264
Toncini G, Barni S, Rescaldani R (1990) Analysis of T helper and suppressor lymphocyte subsets in relation to the clinical stage of solid neoplasms. Oncology 47: 381
Webster DJT, Richardson G, et al (1979) Effect of treatment on the immunological status of women with advanced breast cancer. Br J Cancer 39: 676
White D, Jone DB, Cooke T, Kirham N (1982) Natural killer activity in peripheral blood lymphocytes of patients with benign and malignant disease. Br J Cancer 46: 611
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Robinson, E., Rubin, D., Mekori, T. et al. In vivo modulation of natural killer cell activity by tamoxifen in patients with bilateral primary breast cancer. Cancer Immunol Immunother 37, 209–212 (1993). https://doi.org/10.1007/BF01525437
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01525437