Abstract
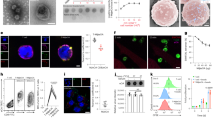
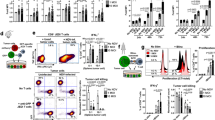
Reovirus is a naturally occurring oncolytic virus currently in early clinical trials. However, the rapid induction of neutralizing antibodies represents a major obstacle to successful systemic delivery. This study addresses, for the first time, the ability of cellular carriers in the form of T cells and dendritic cells (DC) to protect reovirus from systemic neutralization. In addition, the ability of these cellular carriers to manipulate the subsequent balance of anti-viral versus anti-tumour immune response is explored. Reovirus, either neat or loaded onto DC or T cells, was delivered intravenously into reovirus-naive or reovirus-immune C57Bl/6 mice bearing lymph node B16tk melanoma metastases. Three and 10 days after treatment, reovirus delivery, carrier cell trafficking, metastatic clearance and priming of anti-tumour/anti-viral immunity were assessed. In naive mice, reovirus delivered either neat or through cell carriage was detectable in the tumour-draining lymph nodes 3 days after treatment, though complete clearance of metastases was only obtained when the virus was delivered on T cells or mature DC (mDC); neat reovirus or loaded immature DC (iDC) gave only partial early tumour clearance. Furthermore, only T cells carrying reovirus generated anti-tumour immune responses and long-term tumour clearance; reovirus-loaded DC, in contrast, generated only an anti-viral immune response. In reovirus-immune mice, however, the results were different. Neat reovirus was completely ineffective as a therapy, whereas mDC—though not iDC—as well as T cells, effectively delivered reovirus to melanoma in vivo for therapy and anti-tumour immune priming. Moreover, mDC were more effective than T cells over a range of viral loads. These data show that systemically administered neat reovirus is not optimal for therapy, and that DC may be an appropriate vehicle for carriage of significant levels of reovirus to tumours. The pre-existing immune status against the virus is critical in determining the balance between anti-viral and anti-tumour immunity elicited when reovirus is delivered by cell carriage, and the viral dose and mode of delivery, as well as the immune status of patients, may profoundly affect the success of any clinical anti-tumour viral therapy. These findings are therefore of direct translational relevance for the future design of clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- DC:
-
dendritic cell
- iDC:
-
immature DC
- mDC:
-
mature DC
- BMDC:
-
bone marrow-derived dendritic cells
- HSVtk:
-
herpes simplex virus thymidine kinase
- MOI:
-
multiplicity of infection
- TAA:
-
tumour-associated antigens
- T(reo)/DC(reo):
-
T cells/DC loaded with reovirus
- TDLN:
-
tumour-draining lymph nodes
- VSV:
-
vesicular stomatitis virus.
References
Aghi M, Martuza RL . Oncolytic viral therapies—the clinical experience. Oncogene 2005; 24: 7802–7816.
Liu TC, Kirn D . Systemic efficacy with oncolytic virus therapeutics: clinical proof-of-concept and future directions. Cancer Res 2007; 67: 429–432.
Pizzato M, Marlow SA, Blair ED, Takeuchi Y . Initial binding of murine leukemia virus particles to cells does not require specific Env-receptor interaction. J Virol 1999; 73: 8599–8611.
Lyons M, Onion D, Green NK, Aslan K, Rajaratnam R, Bazan-Peregrino M et al. Adenovirus type 5 interactions with human blood cells may compromise systemic delivery. Mol Ther 2006; 14: 118–128.
Worgall S, Wolff G, Falck-Pedersen E, Crystal RG . Innate immune mechanisms dominate elimination of adenoviral vectors following in vivo administration. Hum Gene Ther 1997; 8: 37–44.
Ye X, Jerebtsova M, Ray PE . Liver bypass significantly increases the transduction efficiency of recombinant adenoviral vectors in the lung, intestine, and kidney. Hum Gene Ther 2000; 11: 621–627.
Devaux P, Christiansen D, Plumet S, Gerlier D . Cell surface activation of the alternative complement pathway by the fusion protein of measles virus. J Gen Virol 2004; 85: 1665–1673.
Wakimoto H, Ikeda K, Abe T, Ichikawa T, Hochberg FH, Ezekowitz RA et al. The complement response against an oncolytic virus is species-specific in its activation pathways. Mol Ther 2002; 5: 275–282.
Tsai V, Johnson DE, Rahman A, Wen SF, LaFace D, Philopena J et al. Impact of human neutralizing antibodies on antitumor efficacy of an oncolytic adenovirus in a murine model. Clin Cancer Res 2004; 10: 7199–7206.
Chen Y, Yu DC, Charlton D, Henderson DR . Pre-existent adenovirus antibody inhibits systemic toxicity and antitumor activity of CN706 in the nude mouse LNCaP xenograft model: implications and proposals for human therapy. Hum Gene Ther 2000; 11: 1553–1567.
Fisher K . Striking out at disseminated metastases: the systemic delivery of oncolytic viruses. Curr Opin Mol Ther 2006; 8: 301–313.
Croyle MA, Chirmule N, Zhang Y, Wilson JM . PEGylation of E1-deleted adenovirus vectors allows significant gene expression on readministration to liver. Hum Gene Ther 2002; 13: 1887–1900.
Green NK, Herbert CW, Hale SJ, Hale AB, Mautner V, Harkins R et al. Extended plasma circulation time and decreased toxicity of polymer-coated adenovirus. Gene Therapy 2004; 11: 1256–1263.
Bangari DS, Mittal SK . Current strategies and future directions for eluding adenoviral vector immunity. Curr Gene Ther 2006; 6: 215–226.
Bian H, Fournier P, Moormann R, Peeters B, Schirrmacher V . Selective gene transfer to tumor cells by recombinant Newcastle Disease Virus via a bispecific fusion protein. Int J Oncol 2005; 26: 431–439.
Qiao J, Kottke T, Willmon C, Galivo F, Wongthida P, Diaz RM et al. Purging metastases in lymphoid organs using a combination of antigen-nonspecific adoptive T cell therapy, oncolytic virotherapy and immunotherapy. Nat Med 2008; 14: 37–44.
Iankov ID, Blechacz B, Liu C, Schmeckpeper JD, Tarara JE, Federspiel MJ et al. Infected cell carriers: a new strategy for systemic delivery of oncolytic measles viruses in cancer virotherapy. Mol Ther 2007; 15: 114–122.
Power AT, Wang J, Falls TJ, Paterson JM, Parato KA, Lichty BD et al. Carrier cell-based delivery of an oncolytic virus circumvents antiviral immunity. Mol Ther 2007; 15: 123–130.
Thorne SH, Contag CH . Integrating the biological characteristics of oncolytic viruses and immune cells can optimize therapeutic benefits of cell-based delivery. Gene Therapy 2008; 15: 753–758.
Qiao J, Wang H, Kottke T, Diaz RM, Willmon C, Hudacek A et al. Loading of oncolytic vesicular stomatitis virus onto antigen-specific T cells enhances the efficacy of adoptive T-cell therapy of tumors. Gene Therapy 2008; 15: 604–616.
Thorne SH, Negrin RS, Contag CH . Synergistic antitumor effects of immune cell-viral biotherapy. Science 2006; 311: 1780–1784.
Prestwich RJ, Harrington KJ, Pandha HS, Vile RG, Melcher AA, Errington F . Oncolytic viruses: a novel form of immunotherapy. Expert Rev Anticancer Ther 2008; 8: 1581–1588.
Comins C, Heinemann L, Harrington K, Melcher A, De Bono J, Pandha H . Reovirus: viral therapy for cancer ‘as nature intended’. Clin Oncol (R Coll Radiol) 2008; 20: 548–554.
Coffey MC, Strong JE, Forsyth PA, Lee PW . Reovirus therapy of tumors with activated Ras pathway. Science 1998; 282: 1332–1334.
Tai JH, Williams JV, Edwards KM, Wright PF, Crowe Jr JE, Dermody TS . Prevalence of reovirus-specific antibodies in young children in Nashville, Tennessee. J Infect Dis 2005; 191: 1221–1224.
White CL, Twigger KR, Vidal L, De Bono JS, Coffey M, Heinemann L et al. Characterization of the adaptive and innate immune response to intravenous oncolytic reovirus (Dearing type 3) during a phase I clinical trial. Gene Therapy 2008; 15: 911–920.
Errington F, Steele L, Prestwich R, Harrington KJ, Pandha HS, Vidal L et al. Reovirus activates human dendritic cells to promote innate antitumor immunity. J Immunol 2008; 180: 6018–6026.
Prestwich RJ, Errington F, Ilett EJ, Morgan RS, Scott KJ, Kottke T et al. Tumor infection by oncolytic reovirus primes adaptive antitumor immunity. Clin Cancer Res 2008; 14: 7358–7366.
Cole C, Qiao J, Kottke T, Diaz RM, Ahmed A, Sanchez-Perez L et al. Tumor-targeted, systemic delivery of therapeutic viral vectors using hitchhiking on antigen-specific T cells. Nat Med 2005; 11: 1073–1081.
Errington F, White CL, Twigger KR, Rose A, Scott K, Steele L et al. Inflammatory tumour cell killing by oncolytic reovirus for the treatment of melanoma. Gene Ther 2008; 15: 1257–1270.
Qiao J, Wang H, Kottke T, White C, Twigger K, Diaz RM et al. Cyclophosphamide facilitates antitumor efficacy against subcutaneous tumors following intravenous delivery of reovirus. Clin Cancer Res 2008; 14: 259–269.
Huang J, Tatsumi T, Pizzoferrato E, Vujanovic N, Storkus WJ . Nitric oxide sensitizes tumor cells to dendritic cell-mediated apoptosis, uptake, and cross-presentation. Cancer Res 2005; 65: 8461–8470.
Diaz RM, Galivo F, Kottke T, Wongthida P, Qiao J, Thompson J et al. Oncolytic immunovirotherapy for melanoma using vesicular stomatitis virus. Cancer Res 2007; 67: 2840–2848.
Gunzer M, Janich S, Varga G, Grabbe S . Dendritic cells and tumor immunity. Semin Immunol 2001; 13: 291–302.
Power AT, Bell JC . Cell-based delivery of oncolytic viruses: a new strategic alliance for a biological strike against cancer. Mol Ther 2007; 15: 660–665.
Martin-Fontecha A, Thomsen LL, Brett S, Gerard C, Lipp M, Lanzavecchia A et al. Induced recruitment of NK cells to lymph nodes provides IFN-gamma for T(H)1 priming. Nat Immunol 2004; 5: 1260–1265.
Sivori S, Falco M, Della Chiesa M, Carlomagno S, Vitale M, Moretta L et al. CpG and double-stranded RNA trigger human NK cells by Toll-like receptors: induction of cytokine release and cytotoxicity against tumors and dendritic cells. Proc Natl Acad Sci USA 2004; 101: 10116–10121.
Vidal L, Pandha HS, Yap TA, White CL, Twigger K, Vile RG et al. A phase I study of intravenous oncolytic reovirus type 3 dearing in patients with advanced cancer. Clin Cancer Res 2008; 14: 7127–7137.
Vile RG, Hart IR . Use of tissue-specific expression of the Herpes Simplex virus thymidine kinase gene to inhibit growth of established murine melanomas following direct intratumoural injection of DNA. Cancer Res 1993; 53: 3860–3864.
Melcher A, Todryk S, Bateman A, Chong H, Lemoine NR, Vile RG . Adoptive transfer of immature dendritic cells with autologous or allogeneic tumor cells generates systemic antitumor immunity. Cancer Res 1999; 59: 2802–2805.
Merrick A, Diaz RM, O’Donnell D, Selby P, Vile R, Melcher A . Autologous versus allogeneic peptide-pulsed dendritic cells for anti-tumour vaccination: expression of allogeneic MHC supports activation of antigen specific T cells, but impairs early naive cytotoxic priming and anti-tumour therapy. Cancer Immunol Immunother 2008; 57: 897–906.
Gallucci S, Lolkema M, Matzinger P . Natural adjuvants: endogenous activators of dendritic cells. Nat Med 1999; 5: 1249–1255.
Acknowledgements
We thank Oncolytics Biotech (http://www.oncolyticsbiotech.com) for providing reovirus. This work was funded by Cancer Research UK, and by the Richard M. Schulze Family Foundation, the Mayo Foundation and by NIH Grants CA RO1107082-02 and RO1130878.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ilett, E., Prestwich, R., Kottke, T. et al. Dendritic cells and T cells deliver oncolytic reovirus for tumour killing despite pre-existing anti-viral immunity. Gene Ther 16, 689–699 (2009). https://doi.org/10.1038/gt.2009.29
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2009.29
Keywords
This article is cited by
-
Narrative review on century of respiratory pandemics from Spanish flu to COVID-19 and impact of nanotechnology on COVID-19 diagnosis and immune system boosting
Virology Journal (2022)
-
Improving antitumor efficacy via combinatorial regimens of oncolytic virotherapy
Molecular Cancer (2020)
-
Mesenchymal stem cells used as carrier cells of oncolytic adenovirus results in enhanced oncolytic virotherapy
Scientific Reports (2020)
-
Plasmacytoid dendritic cells orchestrate innate and adaptive anti-tumor immunity induced by oncolytic coxsackievirus A21
Journal for ImmunoTherapy of Cancer (2019)
-
Prime-boost using separate oncolytic viruses in combination with checkpoint blockade improves anti-tumour therapy
Gene Therapy (2017)