Abstract
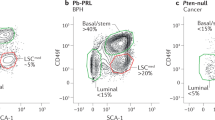
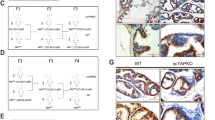
In epithelial tissues, the lineage relationship between normal progenitor cells and cell type(s) of origin for cancer has been poorly understood. Here we show that a known regulator of prostate epithelial differentiation, the homeobox gene Nkx3-1, marks a stem cell population that functions during prostate regeneration. Genetic lineage-marking demonstrates that rare luminal cells that express Nkx3-1 in the absence of testicular androgens (castration-resistant Nkx3-1-expressing cells, CARNs) are bipotential and can self-renew in vivo, and single-cell transplantation assays show that CARNs can reconstitute prostate ducts in renal grafts. Functional assays of Nkx3-1 mutant mice in serial prostate regeneration suggest that Nkx3-1 is required for stem cell maintenance. Furthermore, targeted deletion of the Pten tumour suppressor gene in CARNs results in rapid carcinoma formation after androgen-mediated regeneration. These observations indicate that CARNs represent a new luminal stem cell population that is an efficient target for oncogenic transformation in prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Abate-Shen, C. & Shen, M. M. Molecular genetics of prostate cancer. Genes Dev. 14, 2410–2434 (2000)
English, H. F., Santen, R. J. & Isaacs, J. T. Response of glandular versus basal rat ventral prostatic epithelial cells to androgen withdrawal and replacement. Prostate 11, 229–242 (1987)
Evans, G. S. & Chandler, J. A. Cell proliferation studies in the rat prostate: II. The effects of castration and androgen-induced regeneration upon basal and secretory cell proliferation. Prostate 11, 339–351 (1987)
Sugimura, Y., Cunha, G. R. & Donjacour, A. A. Morphological and histological study of castration-induced degeneration and androgen-induced regeneration in the mouse prostate. Biol. Reprod. 34, 973–983 (1986)
Isaacs, J. T. in Benign Prostatic Hyperplasia (eds Rodgers C. H. et al.) 85–94 (Department of Health and Human Services, 1985)
Tsujimura, A. et al. Proximal location of mouse prostate epithelial stem cells: a model of prostatic homeostasis. J. Cell Biol. 157, 1257–1265 (2002)
Lawson, D. A. & Witte, O. N. Stem cells in prostate cancer initiation and progression. J. Clin. Invest. 117, 2044–2050 (2007)
Senoo, M., Pinto, F., Crum, C. P. & McKeon, F. p63 is essential for the proliferative potential of stem cells in stratified epithelia. Cell 129, 523–536 (2007)
Lawson, D. A., Xin, L., Lukacs, R. U., Cheng, D. & Witte, O. N. Isolation and functional characterization of murine prostate stem cells. Proc. Natl Acad. Sci. USA 104, 181–186 (2007)
Richardson, G. D. et al. CD133, a novel marker for human prostatic epithelial stem cells. J. Cell Sci. 117, 3539–3545 (2004)
Burger, P. E. et al. Sca-1 expression identifies stem cells in the proximal region of prostatic ducts with high capacity to reconstitute prostatic tissue. Proc. Natl Acad. Sci. USA 102, 7180–7185 (2005)
Xin, L., Lawson, D. A. & Witte, O. N. The Sca-1 cell surface marker enriches for a prostate-regenerating cell subpopulation that can initiate prostate tumorigenesis. Proc. Natl Acad. Sci. USA 102, 6942–6947 (2005)
Goldstein, A. S. et al. Trop2 identifies a subpopulation of murine and human prostate basal cells with stem cell characteristics. Proc. Natl Acad. Sci. USA 105, 20882–20887 (2008)
Leong, K. G., Wang, B. E., Johnson, L. & Gao, W. Q. Generation of a prostate from a single adult stem cell. Nature 456, 804–808 (2008)
Kurita, T., Medina, R. T., Mills, A. A. & Cunha, G. R. Role of p63 and basal cells in the prostate. Development 131, 4955–4964 (2004)
Kasper, S. Stem cells: the root of prostate cancer? J. Cell. Physiol. 216, 332–336 (2008)
Wang, S. et al. Pten deletion leads to the expansion of a prostatic stem/progenitor cell subpopulation and tumor initiation. Proc. Natl Acad. Sci. USA 103, 1480–1485 (2006)
Grisanzio, C. & Signoretti, S. p63 in prostate biology and pathology. J. Cell. Biochem. 103, 1354–1368 (2008)
Humphrey, P. A. Diagnosis of adenocarcinoma in prostate needle biopsy tissue. J. Clin. Pathol. 60, 35–42 (2007)
Abate-Shen, C., Shen, M. M. & Gelmann, E. Integrating differentiation and cancer: the Nkx3. 1 homeobox gene in prostate organogenesis and carcinogenesis. Differentiation 76, 717–727 (2008)
Bhatia-Gaur, R. et al. Roles for Nkx3. 1 in prostate development and cancer. Genes Dev. 13, 966–977 (1999)
Abdulkadir, S. A. et al. Conditional loss of Nkx3. 1 in adult mice induces prostatic intraepithelial neoplasia. Mol. Cell. Biol. 22, 1495–1503 (2002)
Kim, M. J. et al. Nkx3. 1 mutant mice recapitulate early stages of prostate carcinogenesis. Cancer Res. 62, 2999–3004 (2002)
Chen, H., Mutton, L. N., Prins, G. S. & Bieberich, C. J. Distinct regulatory elements mediate the dynamic expression pattern of Nkx3. 1 . Dev. Dyn. 234, 961–973 (2005)
Sciavolino, P. J. et al. Tissue-specific expression of murine Nkx3. 1 in the male urogenital system. Dev. Dyn. 209, 127–138 (1997)
Bieberich, C. J., Fujita, K., He, W.-W. & Jay, G. Prostate-specific and androgen-dependent expression of a novel homeobox gene. J. Biol. Chem. 271, 31779–31782 (1996)
Feil, R., Wagner, J., Metzger, D. & Chambon, P. Regulation of Cre recombinase activity by mutated estrogen receptor ligand-binding domains. Biochem. Biophys. Res. Commun. 237, 752–757 (1997)
Indra, A. K. et al. Temporally-controlled site-specific mutagenesis in the basal layer of the epidermis: comparison of the recombinase activity of the tamoxifen-inducible Cre-ER(T) and Cre-ER(T2) recombinases. Nucleic Acids Res. 27, 4324–4327 (1999)
Srinivas, S. et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 1, 4 (2001)
Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nature Genet. 21, 70–71 (1999)
Cunha, G. R. & Vanderslice, K. D. Identification in histological sections of species origin of cells from mouse, rat and human. Stain Technol. 59, 7–12 (1984)
Kiel, M. J. et al. Haematopoietic stem cells do not asymmetrically segregate chromosomes or retain BrdU. Nature 449, 238–242 (2007)
Bickenbach, J. R. & Holbrook, K. A. Label-retaining cells in human embryonic and fetal epidermis. J. Invest. Dermatol. 88, 42–46 (1987)
Cotsarelis, G., Cheng, S. Z., Dong, G., Sun, T. T. & Lavker, R. M. Existence of slow-cycling limbal epithelial basal cells that can be preferentially stimulated to proliferate: implications on epithelial stem cells. Cell 57, 201–209 (1989)
Groszer, M. et al. Negative regulation of neural stem/progenitor cell proliferation by the Pten tumor suppressor gene in vivo . Science 294, 2186–2189 (2001)
Nakagawa, T., Nabeshima, Y. & Yoshida, S. Functional identification of the actual and potential stem cell compartments in mouse spermatogenesis. Dev. Cell 12, 195–206 (2007)
Xu, X. et al. Beta cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell 132, 197–207 (2008)
Barroca, V. et al. Mouse differentiating spermatogonia can generate germinal stem cells in vivo . Nature Cell Biol. 11, 190–196 (2009)
Weaver, M. & Krasnow, M. A. Dual origin of tissue-specific progenitor cells in Drosophila tracheal remodeling. Science 321, 1496–1499 (2008)
Magee, J. A., Abdulkadir, S. A. & Milbrandt, J. Haploinsufficiency at the Nkx3. 1 locus: a paradigm for stochastic, dosage-sensitive gene regulation during tumor initiation. Cancer Cell 3, 273–283 (2003)
Lei, Q. et al. NKX3.1 stabilizes p53, inhibits AKT activation, and blocks prostate cancer initiation caused by PTEN loss. Cancer Cell 9, 367–378 (2006)
Ouyang, X., DeWeese, T. L., Nelson, W. G. & Abate-Shen, C. Loss-of-function of Nkx3. 1 promotes increased oxidative damage in prostate carcinogenesis. Cancer Res. 65, 6773–6779 (2005)
Kim, C. F. et al. Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 121, 823–835 (2005)
Barker, N. et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457, 608–611 (2009)
Banach-Petrosky, W. et al. Prolonged exposure to reduced levels of androgen accelerates prostate cancer progression in Nkx3. 1; Pten mutant mice. Cancer Res. 67, 9089–9096 (2007)
Nagy, A., Gertsenstein, M., Vintersten, K. & Behringer, R. Manipulating the Mouse Embryo: A Laboratory Manual Chs 8–11 359–506 (Cold Spring Harbor Laboratory Press, 2003)
Bunting, M., Bernstein, K. E., Greer, J. M., Capecchi, M. R. & Thomas, K. R. Targeting genes for self-excision in the germ line. Genes Dev. 13, 1524–1528 (1999)
Tybulewicz, V. L., Crawford, C. E., Jackson, P. K., Bronson, R. T. & Mulligan, R. C. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell 65, 1153–1163 (1991)
Deng, C., Wynshaw-Boris, A., Zhou, F., Kuo, A. & Leder, P. Fibroblast growth factor receptor 3 is a negative regulator of bone growth. Cell 84, 911–921 (1996)
Gao, H., Ouyang, X., Banach-Petrosky, W. A., Shen, M. M. & Abate-Shen, C. Emergence of androgen independence at early stages of prostate cancer progression in Nkx3. 1; Pten mice. Cancer Res. 66, 7929–7933 (2006)
Acknowledgements
We thank M. Kim for her initial observations on Nkx3-1 expression in the regressed prostate, and C. Cordon-Cardo, E. Gelmann, C. Mendelsohn and B. Reizis for comments on the manuscript. We are also grateful to C. Bieberich, M. Capecchi, P. Chambon and F. Costantini for providing mice and reagents. This work was supported by grants from the NIH (C.A.-S. and M.M.S.), DOD Prostate Cancer Research Program (K.D.E., C.A.-S. and M.M.S.), and the NCI Mouse Models of Human Cancer Consortium.
Author Contributions X.W., M.K.-D., K.D.E., C.A.-S. and M.M.S. designed experiments, Y.P.-H. and S.M.P. generated mouse reagents, X.W., M.K.-D., K.D.E., D.W., H.Y. and M.V.H. performed experiments, and X.W., M.K.-D., C.A.-S. and M.M.S. wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-5, Supplementary References and Supplementary Figures 1- 9 with Legends. (PDF 2647 kb)
Rights and permissions
About this article
Cite this article
Wang, X., Julio, Md., Economides, K. et al. A luminal epithelial stem cell that is a cell of origin for prostate cancer. Nature 461, 495–500 (2009). https://doi.org/10.1038/nature08361
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08361
This article is cited by
-
Targeting the key players of phenotypic plasticity in cancer cells by phytochemicals
Cancer and Metastasis Reviews (2024)
-
Comprehensive Review on the Effect of Stem Cells in Cancer Progression
Current Tissue Microenvironment Reports (2024)
-
N7-methylguanosine methylation of tRNAs regulates survival to stress in cancer
Oncogene (2023)
-
Experimental in vitro, ex vivo and in vivo models in prostate cancer research
Nature Reviews Urology (2023)
-
Basal stem cell progeny establish their apical surface in a junctional niche during turnover of an adult barrier epithelium
Nature Cell Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.