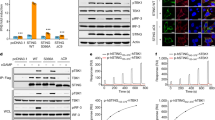
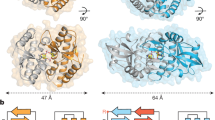
Abstract
Detection of cytoplasmic DNA represents one of the most fundamental mechanisms of the innate immune system to sense the presence of microbial pathogens1. Moreover, erroneous detection of endogenous DNA by the same sensing mechanisms has an important pathophysiological role in certain sterile inflammatory conditions2,3. The endoplasmic-reticulum-resident protein STING is critically required for the initiation of type I interferon signalling upon detection of cytosolic DNA of both exogenous and endogenous origin4,5,6,7,8. Next to its pivotal role in DNA sensing, STING also serves as a direct receptor for the detection of cyclic dinucleotides, which function as second messenger molecules in bacteria9,10,11,12,13. DNA recognition, however, is triggered in an indirect fashion that depends on a recently characterized cytoplasmic nucleotidyl transferase, termed cGAMP synthase (cGAS), which upon interaction with DNA synthesizes a dinucleotide molecule that in turn binds to and activates STING14,15. We here show in vivo and in vitro that the cGAS-catalysed reaction product is distinct from previously characterized cyclic dinucleotides. Using a combinatorial approach based on mass spectrometry, enzymatic digestion, NMR analysis and chemical synthesis we demonstrate that cGAS produces a cyclic GMP-AMP dinucleotide, which comprises a 2′-5′ and a 3′-5′ phosphodiester linkage >Gp(2′-5′)Ap(3′-5′)>. We found that the presence of this 2′-5′ linkage was required to exert potent activation of human STING. Moreover, we show that cGAS first catalyses the synthesis of a linear 2′-5′-linked dinucleotide, which is then subject to cGAS-dependent cyclization in a second step through a 3′-5′ phosphodiester linkage. This 13-membered ring structure defines a novel class of second messenger molecules, extending the family of 2′-5′-linked antiviral biomolecules.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hornung, V. & Latz, E. Intracellular DNA recognition. Nature Rev. Immunol. 10, 123–130 (2010)
Gall, A. et al. Autoimmunity initiates in nonhematopoietic cells and progresses via lymphocytes in an interferon-dependent autoimmune disease. Immunity 36, 120–131 (2012)
Ahn, J., Gutman, D., Saijo, S. & Barber, G. N. STING manifests self DNA-dependent inflammatory disease. Proc. Natl Acad. Sci. USA 109, 19386–19391 (2012)
Ishikawa, H. & Barber, G. N. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 455, 674–678 (2008)
Zhong, B. et al. The adaptor protein MITA links virus-sensing receptors to IRF3 transcription factor activation. Immunity 29, 538–550 (2008)
Jin, L. et al. MPYS, a novel membrane tetraspanner, is associated with major histocompatibility complex class II and mediates transduction of apoptotic signals. Mol. Cell. Biol. 28, 5014–5026 (2008)
Sun, W. et al. ERIS, an endoplasmic reticulum IFN stimulator, activates innate immune signaling through dimerization. Proc. Natl Acad. Sci. USA 106, 8653–8658 (2009)
Ishikawa, H., Ma, Z. & Barber, G. N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 461, 788–792 (2009)
Burdette, D. L. et al. STING is a direct innate immune sensor of cyclic di-GMP. Nature 478, 515–518 (2011)
Huang, Y. H., Liu, X. Y., Du, X. X., Jiang, Z. F. & Su, X. D. The structural basis for the sensing and binding of cyclic di-GMP by STING. Nature Struct. Mol. Biol. 19, 728–730 (2012)
Ouyang, S. et al. Structural analysis of the STING adaptor protein reveals a hydrophobic dimer interface and mode of cyclic di-GMP binding. Immunity 36, 1073–1086 (2012)
Shang, G. et al. Crystal structures of STING protein reveal basis for recognition of cyclic di-GMP. Nature Struct. Mol. Biol. 19, 725–727 (2012)
Shu, C., Yi, G., Watts, T., Kao, C. C. & Li, P. Structure of STING bound to cyclic di-GMP reveals the mechanism of cyclic dinucleotide recognition by the immune system. Nature Struct. Mol. Biol. 19, 722–724 (2012)
Wu, J. et al. Cyclic GMP-AMP is an endogenous second messenger in innate immune signaling by cytosolic DNA. Science 339, 826–830 (2013)
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z. J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013)
Cavlar, T., Deimling, T., Ablasser, A., Hopfner, K. P. & Hornung, V. Species-specific detection of the antiviral small-molecule compound CMA by STING. EMBO J. 32, 1440–1450 (2013)
Rao, F. et al. Enzymatic synthesis of c-di-GMP using a thermophilic diguanylate cyclase. Anal. Biochem. 389, 138–142 (2009)
Davies, B. W., Bogard, R. W., Young, T. S. & Mekalanos, J. J. Coordinated regulation of accessory genetic elements produces cyclic di-nucleotides for V. cholerae virulence. Cell 149, 358–370 (2012)
Conlon, J. et al. Mouse, but not human STING, binds and signals in response to the vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid. J. Immunol. 190, 5216–5225 (2013)
Kristiansen, H., Gad, H. H., Eskildsen-Larsen, S., Despres, P. & Hartmann, R. The oligoadenylate synthetase family: an ancient protein family with multiple antiviral activities. J. Interferon Cytokine Res. 31, 41–47 (2011)
Gao, P. et al. Cyclic [G(2′,5′)pA(3′,5′)p] is the metazoan second messenger produced by DNA-activated cyclic GMP-AMP synthase. Cell 153, 1094–1107 (2013)
Diner, E. J. The innate immune DNA sensory cGAS produces a noncanonical cyclic dinucleotide that activates human STING. Cell Rep. 3, 1355–1361 (2013)
Mizushima, S. & Nagata, S. pEF-BOS, a powerful mammalian expression vector. Nucleic Acids Res. 18, 5322 (1990)
Gaffney, B. L., Veliath, E., Zhao, J. & Jones, R. A. One-flask syntheses of c-di-GMP and the [Rp,Rp] and [Rp,Sp] thiophosphate analogues. Org. Lett. 12, 3269–3271 (2010)
Acknowledgements
We thank M. Pelegrin for providing us with LL171 cells. K.-P.H. is supported by the National Institutes of Health (U19AI083025), the European Research Council Advanced Grant 322869, and the Center for Integrated Protein Science Munich (CIPSM). A.A. and V.H. are members of the excellence cluster ImmunoSensation. V.H. is supported by grants from the German Research Foundation (SFB670) and the European Research Council (ERC 243046).
Author information
Authors and Affiliations
Contributions
A.A., M.G., T.C., G.W., T.D., I.R., J.L., K.-P.H. and V.H. designed experiments and analysed the data. A.A., M.G., T.C., G.W., T.D. and I.R. performed experiments. A.A. and V.H. wrote the manuscript. V.H. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1–15, Supplementary Notes 1–3 and additional references. (PDF 2047 kb)
Rights and permissions
About this article
Cite this article
Ablasser, A., Goldeck, M., Cavlar, T. et al. cGAS produces a 2′-5′-linked cyclic dinucleotide second messenger that activates STING. Nature 498, 380–384 (2013). https://doi.org/10.1038/nature12306
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12306
This article is cited by
-
Cytosolic mtDNA–cGAS–STING axis contributes to sepsis-induced acute kidney injury via activating the NLRP3 inflammasome
Clinical and Experimental Nephrology (2024)
-
Candida albicans extracellular vesicles trigger type I IFN signalling via cGAS and STING
Nature Microbiology (2024)
-
Epstein-Barr virus infection: the micro and macro worlds
Virology Journal (2023)
-
Chronic heat stress induces renal fibrosis and mitochondrial dysfunction in laying hens
Journal of Animal Science and Biotechnology (2023)
-
Role of the cGAS-STING pathway in radiotherapy for non-small cell lung cancer
Radiation Oncology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.