Abstract
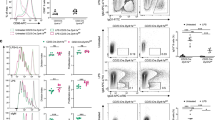
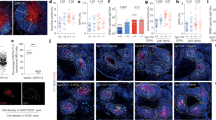
The proliferation of antigen-specific lymphocytes and resulting clonal expansion are essential for adaptive immunity. We report here that B cell–specific deletion of the heavy chain of CD98 (CD98hc) resulted in lower antibody responses due to total suppression of B cell proliferation and subsequent plasma cell formation. Deletion of CD98hc did not impair early B cell activation but did inhibit later activation of the mitogen-activated protein kinase Erk1/2 and downregulation of the cell cycle inhibitor p27. Reconstitution of CD98hc-deficient B cells with CD98hc mutants showed that the integrin-binding domain of CD98hc was required for B cell proliferation but that the amino acid–transport function of CD98hc was dispensable for this. Thus, CD98hc supports integrin-dependent rapid proliferation of B cells. We propose that the advantage of adaptive immunity favored the appearance of CD98hc in vertebrates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Burnet, F.M. The Clonal Selection Theory of Acquired Immunity 49–68 (Cambridge University Press, Cambridge, 1959).
Cooper, M.D. & Alder, M.N. The evolution of adaptive immune systems. Cell 124, 815–822 (2006).
Kehrl, J.H. & Fauci, A.S. Identification, purification, and characterization of antigen-activated and antigen-specific human B lymphocytes. Trans. Assoc. Am. Physicians 96, 182–187 (1983).
Bertran, J. et al. Stimulation of system y+-like amino acid transport by the heavy chain of human 4F2 surface antigen in Xenopus laevis oocytes. Proc. Natl. Acad. Sci. USA 89, 5606–5610 (1992).
Torrents, D. et al. Identification and characterization of a membrane protein (y+L amino acid transporter-1) that associates with 4F2hc to encode the amino acid transport activity y+L. A candidate gene for lysinuric protein intolerance. J. Biol. Chem. 273, 32437–32445 (1998).
Fenczik, C.A., Sethi, T., Ramos, J.W., Hughes, P.E. & Ginsberg, M.H. Complementation of dominant suppression implicates CD98 in integrin activation. Nature 390, 81–85 (1997).
Feral, C.C. et al. CD98hc (SLC3A2) mediates integrin signaling. Proc. Natl. Acad. Sci. USA 102, 355–360 (2005).
Abraham, R.T. Mammalian target of rapamycin: immunosuppressive drugs uncover a novel pathway of cytokine receptor signaling. Curr. Opin. Immunol. 10, 330–336 (1998).
Mondino, A. & Mueller, D.L. mTOR at the crossroads of T cell proliferation and tolerance. Semin. Immunol. 19, 162–172 (2007).
Uinuk-Ool, T. et al. Lamprey lymphocyte-like cells express homologs of genes involved in immunologically relevant activities of mammalian lymphocytes. Proc. Natl. Acad. Sci. USA 99, 14356–14361 (2002).
Tsumura, H. et al. The targeted disruption of the CD98 gene results in embryonic lethality. Biochem. Biophys. Res. Commun. 308, 847–851 (2003).
Feral, C.C. et al. CD98hc (SLC3A2) participates in fibronectin matrix assembly by mediating integrin signaling. J. Cell Biol. 178, 701–711 (2007).
Rickert, R.C., Roes, J. & Rajewsky, K. B lymphocyte-specific, Cre-mediated mutagenesis in mice. Nucleic Acids Res. 25, 1317–1318 (1997).
Otero, D.C. & Rickert, R.C. CD19 function in early and late B cell development. II. CD19 facilitates the pro-B/pre-B transition. J. Immunol. 171, 5921–5930 (2003).
Brakebusch, C. et al. Beta1 integrin is not essential for hematopoiesis but is necessary for the T cell-dependent IgM antibody response. Immunity 16, 465–477 (2002).
Lu, T.T. & Cyster, J.G. Integrin-mediated long-term B cell retention in the splenic marginal zone. Science 297, 409–412 (2002).
Fenczik, C.A. et al. Distinct domains of CD98hc regulate integrins and amino acid transport. J. Biol. Chem. 276, 8746–8752 (2001).
Tangye, S.G. & Hodgkin, P.D. Divide and conquer: the importance of cell division in regulating B-cell responses. Immunology 112, 509–520 (2004).
Hasbold, J., Corcoran, L.M., Tarlinton, D.M., Tangye, S.G. & Hodgkin, P.D. Evidence from the generation of immunoglobulin G–secreting cells that stochastic mechanisms regulate lymphocyte differentiation. Nat. Immunol. 5, 55–63 (2004).
Freidman, A.W., Diaz, L.A., Jr., Moore, S., Schaller, J. & Fox, D.A. The human 4F2 antigen: evidence for cryptic and noncryptic epitopes and for a role of 4F2 in human T lymphocyte activation. Cell. Immunol. 154, 253–263 (1994).
Diaz, L.A., Jr. et al. Monocyte-dependent regulation of T lymphocyte activation through CD98. Int. Immunol. 9, 1221–1231 (1997).
Fernandez-Herrera, J., Sanchez-Madrid, F. & Diez, A.G. Differential expression of the 4F2 activation antigen on human follicular epithelium in hair cycle. J. Invest. Dermatol. 92, 247–250 (1989).
Zent, R. et al. Class- and splice variant-specific association of CD98 with integrin beta cytoplasmic domains. J. Biol. Chem. 275, 5059–5064 (2000).
Proud, C.G. Amino acids and mTOR signalling in anabolic function. Biochem. Soc. Trans. 35, 1187–1190 (2007).
Hynes, R.O. Integrins: bidirectional, allosteric signaling machines. Cell 110, 673–687 (2002).
Schwartz, M.A. & Assoian, R.K. Integrins and cell proliferation: regulation of cyclin-dependent kinases via cytoplasmic signaling pathways. J. Cell Sci. 114, 2553–2560 (2001).
Motti, M.L. et al. Loss of p27 expression through RAS → BRAF → MAP kinase-dependent pathway in human thyroid carcinomas. Cell Cycle 6, 2817–2825 (2007).
Assoian, R.K. & Schwartz, M.A. Coordinate signaling by integrins and receptor tyrosine kinases in the regulation of G1 phase cell-cycle progression. Curr. Opin. Genet. Dev. 11, 48–53 (2001).
Walker, J.L. & Assoian, R.K. Integrin-dependent signal transduction regulating cyclin D1 expression and G1 phase cell cycle progression. Cancer Metastasis Rev. 24, 383–393 (2005).
Lin, K.B. et al. The rap GTPases regulate B cell morphology, immune-synapse formation, and signaling by particulate B cell receptor ligands. Immunity 28, 75–87 (2008).
Arana, E. et al. Activation of the small GTPase Rac2 via the B cell receptor regulates B cell adhesion and immunological-synapse formation. Immunity 28, 88–99 (2008).
Lammermann, T. et al. Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 453, 51–55 (2008).
Wolniak, K.L., Shinall, S.M. & Waldschmidt, T.J. The germinal center response. Crit. Rev. Immunol. 24, 39–65 (2004).
Warren, A.P. et al. Convergence between CD98 and integrin-mediated T-lymphocyte co-stimulation. Immunology 99, 62–68 (2000).
Shimizu, Y., Rose, D.M. & Ginsberg, M.H. Integrins in the immune system. Adv. Immunol. 72, 325–380 (1999).
Sims, T.N. & Dustin, M.L. The immunological synapse: integrins take the stage. Immunol. Rev. 186, 100–117 (2002).
Cyster, J.G. Homing of antibody secreting cells. Immunol. Rev. 194, 48–60 (2003).
Lo, C.G., Lu, T.T. & Cyster, J.G. Integrin-dependence of lymphocyte entry into the splenic white pulp. J. Exp. Med. 197, 353–361 (2003).
Rose, D.M., Alon, R. & Ginsberg, M.H. Integrin modulation and signaling in leukocyte adhesion and migration. Immunol. Rev. 218, 126–134 (2007).
Abram, C.L. & Lowell, C.A. Convergence of immunoreceptor and integrin signaling. Immunol. Rev. 218, 29–44 (2007).
Batista, F.D. et al. The role of integrins and coreceptors in refining thresholds for B-cell responses. Immunol. Rev. 218, 197–213 (2007).
Roovers, K., Davey, G., Zhu, X., Bottazzi, M.E. & Assoian, R.K. Alpha5beta1 integrin controls cyclin D1 expression by sustaining mitogen-activated protein kinase activity in growth factor-treated cells. Mol. Biol. Cell 10, 3197–3204 (1999).
Fleire, S.J. et al. B cell ligand discrimination through a spreading and contraction response. Science 312, 738–741 (2006).
Prager, G.W., Feral, C.C., Kim, C., Han, J. & Ginsberg, M. H. CD98hc (SLC3a2) interaction with the integrin β subunit cytoplasmic domain mediates adhesive signaling. J. Biol. Chem. 282, 24477–24484 (2007).
Esteban, F. et al. Relationship of 4F2 antigen with local growth and metastatic potential of squamous cell carcinoma of the larynx. Cancer 66, 1493–1498 (1990).
Hara, K., Kudoh, H., Enomoto, T., Hashimoto, Y. & Masuko, T. Malignant transformation of NIH3T3 cells by overexpression of early lymphocyte activation antigen CD98. Biochem. Biophys. Res. Commun. 262, 720–725 (1999).
Henderson, N.C. et al. CD98hc (SLC3A2) interaction with β1 integrins is required for transformation. J. Biol. Chem. 279, 54731–54741 (2004).
White, D.E. et al. Targeted disruption of β1-integrin in a transgenic mouse model of human breast cancer reveals an essential role in mammary tumor induction. Cancer Cell 6, 159–170 (2004).
Kass, L., Erler, J.T., Dembo, M. & Weaver, V.M. Mammary epithelial cell: influence of extracellular matrix composition and organization during development and tumorigenesis. Int. J. Biochem. Cell Biol. 39, 1987–1994 (2007).
Van Parijs, L. et al. Uncoupling IL-2 signals that regulate T cell proliferation, survival, and Fas-mediated activation-induced cell death. Immunity 11, 281–288 (1999).
Acknowledgements
We thank M. Slepak for technical assistance; D. Rose for advice on analyzing antibody responses; M. Cato for advice and protocols for enzyme-linked immunospot assays; and R. Zent (Vanderbilt University) for Slc3a2+/− mice. Supported by the National Institutes of Health (AR27214, HL31950 and HL0780784), the Arthritis Foundation (C.C.F.) and the National Multiple Sclerosis Society (FG1802-A-1 to J.C.).
Author information
Authors and Affiliations
Contributions
M.H.G., J.C. and C.C.F. conceived the project with advice and collaboration from R.C.R.; J.C. and M.H.G. wrote the manuscript with editorial input from R.C.R. and C.C.F.; R.C.R. provided CD19-Cre mice; J.C. did experiments except as follows: C.D.B. did immunohistochemical analysis of spleen sections and flow cytometry for B cell subsets, and R.R. tested B cell proliferation in pan integrin-deficient mice.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13 and Supplementary Methods (PDF 699 kb)
Rights and permissions
About this article
Cite this article
Cantor, J., Browne, C., Ruppert, R. et al. CD98hc facilitates B cell proliferation and adaptive humoral immunity. Nat Immunol 10, 412–419 (2009). https://doi.org/10.1038/ni.1712
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1712
This article is cited by
-
HSV-1 0∆NLS vaccine elicits a robust B lymphocyte response and preserves vision without HSV-1 glycoprotein M or thymidine kinase recognition
Scientific Reports (2022)
-
An anti-CD98 antibody displaying pH-dependent Fc-mediated tumour-specific activity against multiple cancers in CD98-humanized mice
Nature Biomedical Engineering (2022)
-
Oncogenic RAS commandeers amino acid sensing machinery to aberrantly activate mTORC1 in multiple myeloma
Nature Communications (2022)
-
Infection-induced plasmablasts are a nutrient sink that impairs humoral immunity to malaria
Nature Immunology (2020)
-
mTOR intersects antibody-inducing signals from TACI in marginal zone B cells
Nature Communications (2017)