Abstract
Two competing theories have been put forward to explain the role of CD4+ T cells in priming CD8+ memory T cells: one proposes paracrine secretion of interleukin 2 (IL-2); the other proposes the activation of antigen-presenting cells (APCs) via the costimulatory molecule CD40 and its ligand CD40L. We investigated the requirement for IL-2 by the relevant three cell types in vivo and found that CD8+ T cells, rather than CD4+ T cells or dendritic cells (DCs), produced the IL-2 necessary for CD8+ T cell memory. Il2−/− CD4+ T cells were able to provide help only if their ability to transmit signals via CD40L was intact. Our findings reconcile contradictory elements implicit in each model noted above by showing that CD4+ T cells activate APCs through a CD40L-dependent mechanism to enable autocrine production of IL-2 in CD8+ memory T cells.
Similar content being viewed by others
Main
The initial encounter with antigen-presenting cells (APCs) constitutes the primary force behind the population expansion, differentiation and survival of naive T cells. Once set into motion by inductive signals received during this priming event, the development of both effector and memory CD8+ T cells can be guided by an instructional program executed without the need for further antigenic stimulation1,2,3,4,5,6. CD4+ helper T cells have an important role in establishing the components of this program, and their participation can endow CD8+ T cells with a key function of immune memory: the ability to undergo secondary proliferative responses after reencountering antigen7,8,9,10. A crucial aspect of this mechanism involves alteration of the developmental program in 'helped' CD8+ T cells to prevent the activation-induced cell death mediated by TRAIL that occurs after restimulation of CD8+ T cells that have not received help ('helpless'; those primed in the absence of CD4+ T cells)11,12. A long-standing controversy in this process has focused on the sequence and nature of the cellular interactions through which help is provided. Helper T cells have been shown to have a crucial role in licensing APCs, through sequential two-cell interactions involving the costimulatory molecule CD40 and its ligand CD40L, from a resting to an activated state in which they are subsequently able to directly prime CD8+ T cells13,14,15,16 An alternative model suggests that a CD4+ T cell helps a CD8+ T cell via paracrine secretion of the growth and differentiation factor interleukin 2 (IL-2) when both are in a three-cell cluster with an APC17,18,19. Although the three-cell cluster model was initially based on demonstration of the epitope linkage that would be required for CD4+ T cell–CD8+ T cell collaboration and in vitro restimulation studies, more direct in vivo evidence has demonstrated that CD8+ T cells require IL-2 signals during priming to enable their clonal progeny to mount optimal secondary proliferative responses20. Prolonged IL-2 signaling has been shown to promote the development of (short-lived) effector CD8+ T cells, which suggests that the magnitude or duration of IL-2 signals can have a profound influence on the functional differentiation of the cells21,22. Such studies have elucidated the effect of IL-2 on CD8+ T cells but have not provided information about its relevant physiological source in vivo. Here we investigated the cell types known to produce IL-2 in vivo (CD4+ T cells, CD8+ T cells and dendritic cells (DCs)) to identify which are crucial for programming the capacity for the secondary population expansion of memory CD8+ T cells. Our results indicate that CD4+ T cells required the ability to transmit CD40L signals but did not need to produce IL-2 to provide the help needed for CD8+ T cell memory, and that autocrine IL-2 produced by the 'helped' memory CD8+ T cells enabled their optimal secondary population expansion.
Results
Helper T cells require CD40L but not IL-2
The CD4+ T cell–dependent priming of CD8+ T cells involves three distinct types of cells: a CD4+ T cell, a CD8+ T cell and an APC bearing the surface peptide–major histocompatibility complex (MHC) ligands recognized by each. As each of these cell types has the ability to produce IL-2, we sought to establish which functions as the relevant source of this cytokine in vivo to enable the secondary proliferative response of CD8+ T cells. Studies of mice lacking expression of IL-2 or its receptor CD25 (IL-2 receptor α-chain) have been complicated by the fact that these mice suffer from a range of autoimmune and lymphoproliferative T cell disorders presumably due to the lack of IL-2-dependent CD4+ regulatory T cells23,24. To overcome this, we used a strategy in which we specifically controlled the production of IL-2 in vivo in each of the relevant cell types able to produce this cytokine during CD8+ T cell priming. This involved the adoptive transfer of wild-type or Il2−/− CD4+ (OT-II) or CD8+ (OT-I) T cells specific for the model antigen chicken ovalbumin (OVA) into wild-type isogenic hosts, or of OVA-expressing wild-type or Il2−/− DCs that are the only APC able to present the complex of OVA amino acids 257–264 (OVA(257–264)) and H-2Kb recognized in vivo by OT-I CD8+ T cells in H-2Kbm1 hosts (a strain unable to present this peptide to H-2Kb-restricted CD8+ T cells)25.
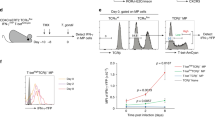
We first investigated whether IL-2 production by CD4+ T cells was required for the priming of CD8+ T cells able to undergo secondary population expansion. To achieve this, we adoptively transferred 5 × 104 wild-type or Il2−/− OT-II T cells into adult mice that had undergone thymectomy and had been depleted of endogenous CD4+ T cells by treatment with monoclonal antibody 15 d before to ensure that any CD4+ T cell help would derive from the transferred cells (Supplementary Fig. 1a). After immunization and subsequent rechallenge, control mice depleted of CD4+ T cells mounted only weak primary and secondary CD8+ T cell responses (as determined by analysis of the OVA(257–264)–H-2Kb tetramer and production of interferon-γ (IFN-γ)), in contrast to the strong responses in those that received either wild-type or Il2−/− OT-II CD4+ T cells (Fig. 1), which confirmed the dependence of CD8+ T cell responses on CD4+ T cells in this experimental system. Notably, we found no great difference in the magnitude of the primary and secondary endogenous antigen-specific CD8+ T cell responses, either by percentage or absolute numbers, in mice that received either wild-type or Il2−/− CD4+ T cells (Fig. 1a–c). The OT-II response nonetheless did include an IL-2-producing component (Supplementary Fig. 1b), and the help provided by CD4+ T cells was clearly antigen specific (Supplementary Fig. 1c). In contrast, antibody blockade of CD40L prevented both wild-type and Il2−/− OT-II cells from providing help to the same endogenous CD8+ T cells (Fig. 1d). These data demonstrate that although CD4+ T cells are critical in priming a robust primary and secondary CD8+ T cell response from endogenous polyclonal precursors, their ability to produce IL-2 is irrelevant to this process. The ability of CD4+ T cells to transmit the 'help message', however, is completely dependent on intact CD40-CD40L interactions, in support of the sequential two-cell interaction model16.
(a) Flow cytometry of splenic CD8+ T cells from C57BL/6J mice that had undergone thymectomy and were first depleted of CD4+ T cells, then given injection of PBS alone (No OT-II) or 5 × 104 OT-II cells (OT-II) or OT-II Il2−/− cells (OT-II Il2−/−), then primed 1 d later with 5 × 106 Act-mOVA H-2Kb-deficient splenocytes and challenged 30 d later with 5 × 106 plaque-forming units (PFU) of vaccinia virus–OVA. Numbers adjacent to outlined areas indicate percent H-2Kb–OVA tetramer–positive CD8+ T cells (mean ± s.e.m. of six mice). (b,c) Frequency (b) and absolute number (c) of IFN-γ+ CD8+ T cells among total splenocytes from the mice in a (n = 6 per group), assessed by intracellular staining at day 7 (primary) or at day 35 in mice that had (secondary) or had not (memory) been challenged with vaccinia virus–OVA 5 d earlier. Each symbol represents an individual mouse; small horizontal lines indicate the mean (b). (d) Flow cytometry of CD8+ T cells in blood from the mice in a that were given no antibody treatment (far left) or treated with control immunoglobulin (control Ig) or blocking antibody to CD40L (α-CD40L), assessed at day 7 (primary), day 30 (memory) or day 35 (secondary) after mice were challenged with vaccinia virus–OVA 5 d earlier. Numbers adjacent to outlined areas indicate percent OVA(257–264)–H-2Kb tetramer–specific CD8+ T cells (mean ± s.e.m. of six mice). NS, not significant; *P < 0.05 (two-tailed unpaired t-test). Data are representative of three (a,b) or two (c,d) independent experiments (mean ± s.e.m.).
DC-derived IL-2 is not essential for CD8+ memory
To determine if IL-2 production by APCs is necessary for CD8+ T cell memory, we used a system in which a population of adoptively transferred DCs serves as the only APCs able to prime CD8+ T cells specific for OVA(257–264)–H-2Kb in vivo. This involved the generation of DCs from wild-type (Il2+/+) or Il2−/− Act-mOVA mice, which express a membrane-bound form of OVA at their cell surface and present OVA-derived peptides on their surface MHC class I and class II molecules26. To ensure that only the transferred DCs were able to present OVA and to avoid cross-presentation, we used H-2Kbm1 mice as recipients because endogenous APCs from this mouse strain are unable to present the OVA(257–264) peptide to H-2Kb-restricted CD8+ T cells such as OT-I cells via the Kbm1 variant form of the H-2Kb MHC molecule they express27. We first gave H-2Kbm1 mice OT-I Kbm1 CD8+ T cells (from mice backcrossed to the H-2Kbm1 strain to prevent rejection in the H-2Kbm1 host) and then immunized the mice 1 d later with wild-type or IL-2-deficient Act-mOVA DCs that had been exposed to the Toll-like receptor 9 ligand CpG to induce IL-2 secretion28. We subsequently monitored the primary and secondary population expansion of OVA-specific CD8+ T cells at day 7 after immunization and then again over 5 months later after challenge with wild-type or IL-2-deficient Act-mOVA DCs that had been activated by CpG. Although the Act-mOVA DCs transferred into the H-2Kbm1 hosts in this system would be expected to be rejected by host T cells because of their expression of a foreign MHC (H-2Kb), this did not preclude their ability to transmit the signals necessary to generate both primary and memory CD8+ T cells (Fig. 2). Notably, we found that the absence of IL-2 on either the priming or boosting APC had no discernable effect on the magnitude of the primary or secondary proliferative response of the OT-I CD8+ T cells. These results demonstrate that DC-derived IL-2 is not required for the programming of CD8+ T cell memory.
(a) Flow cytometry of splenocytes from H-2Kbm1 mice given injection of 5 × 104 OT-I H-2Kbm1 CD45.1+ cells, then primed 1 d later with 7 × 104 wild-type (WT) or Il2−/− Act-mOVA DCs, followed by analysis of the primary CD8+ T cell response after 7 d (top row) or boosted 139 d later with 7 × 105 wild-type or Il2−/− Act-mOVA DCs, followed by analysis of the OT-I response in both primed and boosted groups 5 d later. Numbers adjacent to outlined areas indicate percent OT-I CD8+ T cells among total splenocytes (mean ± s.e.m.). (b) Absolute number of OT-I CD8+ T cells among total splenocytes during the memory and secondary response in a. Data are representative of two experiments (mean ± s.e.m. of three to four mice in b).
CD8+ T cells require autocrine IL-2
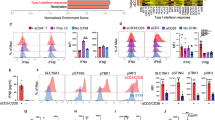
To determine whether the ability to produce autocrine IL-2 is important for CD8+ T cell memory, we monitored the response of a physiological number (∼50) of wild-type versus Il2−/− OT-I CD8+ T cells after adoptive transfer into wild-type mice. This approach allowed us to simultaneously track both wild-type endogenous CD8+ T cells and transferred Il2−/− OT-I CD8+ T cells at each phase (primary, memory and secondary) of a CD4+ T cell–dependent CD8+ T cell response induced by heterologous priming and boosting with infectious pathogens (vaccinia virus–OVA or Listeria monocytogenes–OVA)29. We found that OT-I cells lacking IL-2 expression underwent a more modest primary population expansion than did wild-type OT-I cells, as reported before30, although we reproducibly detected a population of primary memory cells before rechallenge (Fig. 3). After challenge of intact recipient mice, however, the Il2−/− OT-I CD8+ T cells had a profound defect in their secondary population expansion similar in magnitude to that of either endogenous or OT-I CD8+ T cells that had not received help (Fig. 3a–c). The endogenous IL-2-sufficient CD8+ T cells, in contrast, underwent a robust secondary population expansion in the intact recipients (Fig. 3c), which showed that adequate help was indeed provided by host CD4+ T cells in this setting. Despite the availability of CD4+ T cell help, only the CD8+ T cells lacking IL-2 had a defect in their secondary population expansion. Together these data show that under conditions of heterologous priming and boosting with infectious pathogens, the ability to produce autocrine IL-2 is critical for CD8+ T cells to mount optimal secondary proliferative responses.
(a,b) Frequency (a) and absolute number (b) of OT-I (CD45.1+) CD8+ T cells among total splenocytes from intact C57BL/6J (CD45.2+) mice (+ help) or C57BL/6J (CD45.2+) mice depleted of CD4+ T cells (– help; n = 3–4 per group) given 50 wild-type or Il2−/− (CD45.1+) OT-I CD8+ T cells, then infected 1 d later with 1 × 106 PFU vaccinia virus–OVA and, for some groups (secondary), challenged 40 d later with 0.6 half-maximal lethal dose of L. monocytogenes–OVA, followed by analysis at day 7 (primary), or at day 45 for mice that had (secondary) or had not (memory) been challenged with L. monocytogenes–OVA 5 d earlier. Each symbol represents an individual mouse; small horizontal lines indicate the average (a). *P < 0.05 (two-tailed unpaired t-test). Data are representative of three experiments (error bars (b), s.e.m.). (c) IFN-γ production by the endogenous (CD45.1−) and OT-I (CD45.1+) CD8+ T cells during the primary, memory and secondary responses in a,b. Numbers in quadrants indicate percent cells in each (mean ± s.e.m.). Data are representative of three experiments with 8–12 mice. (d) Population expansion of the OT-I CD8+ T cells in a,b. Data are representative of three experiments.
To investigate whether those results were also obtained in response to nonreplicating, noninflammatory immunogens, we immunized C57BL/6 mice containing wild-type or Il2−/− OT-I CD8+ T cells with a cell-based immunogen: Act-mOVA splenocytes with homozygous deletion of the gene encoding H-2Kb. In this setting, the OVA antigen must be cross-presented by host APCs because of the absence of the H-2Kb MHC class I molecule required for direct presentation of the relevant OVA(257–264) peptide to CD8+ T cells31. Although the OVA-specific responses to the cell-based immunogen were, as a rule, smaller than those induced by infectious pathogens, we nonetheless clearly observed the specific defect in the secondary population expansion of Il2−/− CD8+ T cells after rechallenge (Fig. 4). Together these results show that IL-2 produced by CD8+ T cells is essential for their optimal secondary population expansion in response to both replicating and nonreplicating immunogens, even under conditions in which adequate help is available from CD4+ T cells able to produce paracrine IL-2.
(a,b) Frequency (a) and absolute number (b) of OT-I (CD45.1+) CD8+ T cells among total splenocytes from C57BL/6 (CD45.2+) mice (n = 3–4 per group) given 50 wild-type or Il2−/− (CD45.1+) OT-I CD8+ T cells, then immunized 1 d later with 5 × 106 Act-mOVA H-2Kb-deficient splenocytes and, for some groups (secondary), challenged 30 d later with 5 × 106 PFU vaccinia virus–OVA, followed by analysis at day 7 (primary), or at day 45 in mice that had (secondary) or had not (memory) been challenged with vaccinia virus–OVA 5 d (presented as in Fig. 3a,b). (c) IFN-γ production by endogenous (CD45.1−) and OT-I (CD45.1+) CD8+ T cells during the primary, memory and secondary responses in a (presented as in Fig. 3c; n = 8–12 mice). Data are representative of two experiments (mean ± s.e.m. in b,c).
Function and phenotype of Il2−/− CD8+ T cells
Phenotypic examination of wild-type and IL-2-deficient OT-I cells indicated that those lacking IL-2 produced fewer multifunctional cytokine-producing cells (positive for IFN-γ and tumor necrosis factor) during all measured phases of the response to infectious pathogens, with the most profound deficit being apparent in the memory population (Fig. 5a). Monitoring of surface markers showed no substantial difference in the frequency of short-lived effector cells (KLRG-1+CD127−), memory precursor effector cells (KLRG-1−CD127+), central memory cells (CD127+ CD62L+) or effector memory cells (CD62L−CD127+) among wild-type or IL-2-deficient OT-I cells at each phase of the response (Fig. 5b,c). Both wild-type and IL-2-deficient OT-I cells had in vivo cytotoxic ability (Fig. 5d), although the latter cells did show a delay in the production of granzyme B (Fig. 5e). The defect in secondary population expansion in IL-2-deficient OT-I cells was not due to altered migration activity relative to that of wild-type cells, as we found similar ratios of the two populations in every organ assessed (Supplementary Fig. 2). These data show that autocrine IL-2 is not strictly required for cytotoxic function by CD8+ T cells or for their ability to acquire the surface phenotype associated with either central and effector memory cells.
(a) Frequency of OT-I CD8+ T cells producing IFN-γ alone (IFN-γ+) or both IFN-γ and tumor necrosis factor (IFN-γ+TNF+) among total splenocytes from C57BL/6J (CD45.2+) mice (n = 3–4 per group) given wild-type (OT-I) or Il2−/− (OT-I Il2−/−) CD45.1+ OT-I CD8+ T cells and infected 1 d later with 1 × 106 PFU vaccinia virus–OVA, analyzed by intracellular staining of CD45.1+ cells at day 7 (primary), or at day 45 in mice that had (secondary) or had not (memory) been challenged with 0.6 half-maximal lethal dose of L. monocytogenes–OVA 5 d earlier. *P < 0.05 and **P < 0.005 (two-tailed unpaired t-test). (b) Frequency of OT-I cells with positive or negative expression of the activation and memory markers KLRG-1 and CD127 (IL-7 receptor α-chain) on CD45.1+ cells in the blood from the mice in a at day 7 (primary). (c) Frequency of OT-I CD8+ T cells with the surface phenotype of central memory cells (CD127+CD62L+) or effector memory cells (CD127+CD62L−) at day 40 (memory) in the blood of the mice in a. (d) In vivo cytotoxicity of wild-type and Il2−/− OT-I cells 6 d after immunization with vaccinia virus–OVA, presented as the frequency of tetramer-positive OT-I cells in wild-type mice (numbers above outlined areas; left) and frequency of target cells loaded with various concentrations of the cytosolic dye CSFE and pulsed with the peptide epitope of adenovirus early 1B protein amino acids 192–200 (E1B) or OVA(257–264) (OVA) 16 h after adoptive transfer into the immunized mice (right; numbers in plots indicate percent CSFE+ cells). (e) Expression of granzyme B by wild-type and Il2−/− OT-I cells at days 0–5 after immunization of the mice in a with vaccinia virus–OVA. IgG (key), staining with isotype-matched control antibody. Data are representative of three (a,c) or two (b,d,e) experiments (error bars (a–c), s.e.m.).
Discussion
The findings reported here have demonstrated that CD8+ T cells, rather than CD4+ T cells, constitute the cellular source of IL-2 required for the generation of helper T cell–dependent CD8+ T cell memory. The contribution of CD4+ T cells to this process was mediated through their ability to activate APCs via CD40L-CD40 interactions to a state in which they became able to endow the CD8+ T cells they primed with the ability to produce their own autocrine IL-2. Our findings are inconsistent with several tenets of the three-cell model of helper T cell–dependent CD8+ T cell priming, which envisions the nearly contemporaneous interaction of an antigen-specific CD4+ T cell and CD8+ T cell at the surface of the same APC so that paracrine secretion of IL-2 (by the CD4+ T cell for the CD8+ T cell) can take place, and instead support the idea that, when properly stimulated by the appropriate signals emanating from a CD40-activated APC, a CD8+ T cell becomes competent to make its own IL-2. Our findings reconcile discrepancies regarding the specific role of CD4+ T cells and the source of IL-2 required for optimal secondary population expansion inherent in the three-cell model versus the sequential interaction model and allow a meaningful extension of the latter by ascribing IL-2 production to the CD8+ T cell rather than the CD4+ T cell.
Our results are consistent with published findings showing that CD8+ memory T cells (which have low expression of the CD25 (IL-2 receptor α-chain)) have a greater capacity to produce this cytokine21. Furthermore, our results have the potential to inform the finding that Il2−/− CD8+ T cells can undergo secondary proliferation in a bone marrow chimera if provided with paracrine IL-2 (ref. 20). Given that, it is conceivable that in certain model systems, immunogens that induce a T cell response of sufficient magnitude will generate enough paracrine IL-2 at sites of activation to overcome the autocrine requirement demonstrated by our studies. Similarly, the cellular sources for IL-2 required for the population expansion of CD8+ memory T cells may differ depending on the nature of the priming stimulus, the number of each cell type recruited into the response and the microenvironmental localization. Our findings contrast with those of a published study supporting the possibility of a role for paracrine secretion of IL-2 by CD4+ T cells, which might be explained by the nature of the priming stimulus (peptide-loaded DC versus cross-presented antigen) or perhaps in the different numbers of antigen-specific CD4+ T cells used in the two experimental systems32. It is possible that the experimental system used in that study32, which featured the presence of up to 20-fold more OT-II cells during priming than used in our experiments here, may have influenced the quantity of IL-2 produced and thus the distinct results we obtained. We also acknowledge that the structure of secondary lymphoid organs in our system may have been altered as a consequence of adult thymectomy and antibody-mediated depletion of mature CD4+ T cells. Whatever the degree of potential structural alteration, however, it did not preclude the generation of robust CD4+ T cell–dependent OVA-specific memory CD8+ T cells from endogenous responders. Notably, our study has provided evidence that CD4+ T cells do not need to produce IL-2 to provide the help needed for CD8+ T cell memory and confirm in a second model of CD4+ T cell–dependent CD8+ T cell priming that blockade of CD40L prevents transmission of the help signal from CD4+ T cells to APCs13,15,16.
Although IL-2 produced by DCs is important for the initiation of innate immune responses, our results have demonstrated that this did not influence the outcome of the adaptive CD8+ T cell response during either priming or secondary antigenic encounter33. Under certain conditions, such as after immunization with virus or live vector or adjuvant known to recruit DCs and to enhance their persistence at the site of immune-response initiation, the requirement for DC-derived IL-2 may be altered. The H-2Kbm1 model system used in our experiments could prove useful in other settings, as it allows the in vivo antigen-presenting ability of many different cell types to be evaluated. Although the H-2Kbm1 molecule poorly selects the OT-I T cell antigen receptor, we were nonetheless able to isolate fully functional mature OT-I cells from the OT-I.Kbm1 strain for use in adoptive transfer experiments34.
Our studies have identified additional aspects of the mechanism underlying the role of helper T cells in the formation of CD8+ memory T cells, which have been shown to act from the earliest stages of this process in directing the chemokine-mediated attraction of naive CD8+ T cells to sites of interaction between DCs and CD4+ T cells, and, as reinforced by the findings of our study, by activating DCs via CD40L-CD40 interactions so they can transmit the signals necessary to prime CD8+ memory T cells13,15,16,35. Although our studies did not specify a role for helper T cell–derived IL-2 in this process, we must point out that paracrine IL-2 was nonetheless available from either CD4+ T cells or DCs in our experimental systems. Despite that, paracrine IL-2 was not sufficient to 'rescue' the secondary population expansion defect of the Il2−/− CD8+ T cells. Our finding that such cells functioned as cytotoxic effectors in vivo is consistent with results obtained with Il2−/− mice, although our additional information about the presence of considerably fewer cells producing both IFN-γ and tumor necrosis factor may provide new insight into the role of autocrine IL-2 in the generation of the multifunctional subset that bears further investigation36.
The observation that CD4+ T cell–derived IL-2 was irrelevant for the priming of memory CD8+ T cells removes a central function ascribed to these cells in the three-cell model, as well as the requirement for the physical proximity of CD4+ T cells and CD8+ T cells invoked by the paracrine secretion model. By enabling CD8+ T cells to produce and consume their own IL-2, the architecture of the immune system confers a considerable degree of independence on these antigen-experienced 'killer' T cells and enables them to undergo both cytolytic and proliferative responses without requiring their interaction with either antigen-specific CD4+ T cells or professional IL-2-expressing activated APCs, both of which may be either rare or transitory under physiological conditions of re-encountering antigen at peripheral sites of infection. The findings reported here provide new insight into the mechanism through which helper T cells enable CD8+ T cells to achieve the functional autonomy essential for their task as armed sentinels patrolling both lymphoid and peripheral environments. Future efforts should be aimed at identifying the APC-derived signals that 'imprint' CD8+ T cells with the ability to produce autocrine IL-2, determining the point(s) during the development of CD8+ T cell memory at which autocrine IL-2 acts to enable memory cell formation and elucidating whether its main effect is related to its role in promoting metabolism or new patterns of gene expression37.
Methods
Mice.
C57BL/6 mice, B6.C-H2bm1/ByJ (H-2Kbm1) mice, C57BL/6J mice and adult C57BL/6J mice that had undergone thymectomy were from The Jackson Laboratory. Act-mOVA-transgenic mice were a gift from M.K. Jenkins. Il2−/− mice, OT-I recombination-activating gene 1–deficient (Rag1−/−) CD45.1+ mice and Act-mOVA-Kb-deficient mice on the C57BL/6J background have been described11,38. The OT-I Rag1−/−Il2−/− (CD45.1+), OT-II Rag1−/−Il2−/− and Act-mOVA Il2−/− strains were generated by intercrossing. Mice were maintained and bred in the La Jolla Institute for Allergy and Immunology vivarium under specific pathogen–free conditions in accordance with guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care International.
Antibody treatment.
For OT-I T cell responses, 'helpless' groups received intraperitoneal injection of 200 μg antibody to CD4 (anti-CD4; GK1.5; produced 'in-house') 3 d and 1 d before immunization, which resulted in the depletion of >99% of CD4+ T cells, confirmed by staining with phycoerythrin-labeled noncompeting anti-CD4 (RM4.4; eBioscience). Mice in both the 'helped' and 'helpless' groups received 200 μg anti-CD4 (GK1.5) 4 d after primary immunization. For endogenous T cell responses, C57BL/6J adult mice that had undergone thymectomy received an initial dose of 200 μg anti-CD4 (GK1.5) and a second dose of 100 μg 2 d later. Then, 2 weeks after that final dose, cells were stained with anti-CD4 for confirmation of the absence of CD4+ cells (Supplementary Fig. 1a). For CD40L blockade, mice received 150 μg antibody to mouse CD40L (MR-1; produced 'in-house') or isotype-matched polyclonal control hamster immunoglobulin G (007-000-003; Jackson ImmunoResearch) on days −1, 0 and +2 relative to immunization.
T cell preparation.
OT-I (CD45.1+) or OT-I Il2−/− (CD45.1+) cells were collected from blood. Mice received 50 OT-I cells by intravenous injection mixed with 1 × 106 P14 CD90.1+ splenocytes used as an internal control for acquisition by flow cytometry. OT-I Kbm1 cells were collected from the spleen and 5 × 104 cells were injected. OT-II or OT-II Il2−/− CD4+ T cells or CD4+ T cells from SMARTA mice (with transgenic expression of a T cell antigen receptor specific for lymphocytic choriomeningitis virus glycoprotein) were isolated by negative selection (MACS; Miltenyi), and 5 × 104 cells were injected intravenously. OT-I Kbm1 CD45.1+ cells were isolated by positive selection (MACS; Miltenyi). The number of each transferred cell type was determined by direct counting and by flow cytometry staining for the specific combination of T cell antigen receptor α-chain and β-chain variable region they express.
DC preparation.
Offspring produced by intercrossing of Act-mOVA Il2+/− mice were injected between day 5 and day 10 after birth with bone marrow and splenocytes from wild-type CD45.1+ mice to inhibit the lymphoproliferation due to the lack of regulatory T cells in the Il2−/− mice produced in the F1 generation. At 8 weeks of age, mice were implanted subcutaneously with B16 mouse melanoma cells expressing the cytokine Flt3L to increase the number of splenic DCs collected 15 d later39. Spleens from wild-type or Il2−/− Act-mOVA mice were perfused with collagenase D (1 mg/ml) in presence of DNAse I and isolated as described40. DCs were activated for 30 min at 37 °C with 1 mM CpG and stained with allophycocyanin-conjugated anti-CD11c (HL3; BD Pharmingen), phycoerythrin-conjugated anti-CD45.2 (104; eBioscience) and 7-amino-actinomycin D before sorting to obtain CD11c+CD45.2+ cells negative for 7-amino-actinomycin D.
In vivo experiments.
OT-I, OT-II or SMARTA cells were transferred into recipient mice 1 d before immunization. Mice were immunized intraperitoneally with 5 × 106 Act-mOVA H-2Kb-deficient splenocytes (priming) and were challenged intraperitoneally with 5 × 106 PFU vaccinia virus–OVA, or were primed with 1 × 106 PFU vaccinia virus–OVA and boosted intravenously with 3 × 103 colony-forming units of Listeria monocytogenes–OVA. For DC experiments, groups of H-2Kbm1 (CD45.2+) mice initially received 5 × 104 OT-I H-2Kbm1 (CD45.1+) cells and were injected intravenously 1 d later with 5 × 104 Act-mOVA Il2+/+ or Il2−/− DCs. Then, 7 d later, spleens were isolated and the response of the OT-I Kbm1 (CD45.1+) T cells was analyzed by measurement of the frequency of CD45.1+ cytokine-producing cells. Some groups were boosted intravenously with 5 × 105 Act-mOVA Il2+/+ or Il2−/− DC at day 139 after infection and the secondary responses were measured 5 d later.
Ex vivo restimulation and antibody staining.
Cytokine production in splenic CD8+ T cells was assessed with the Fixation/Permeabilization Solution Kit with BD GolgiPlug according to the manufacturer's instructions (BD Pharmingen) after in vitro restimulation for 5 h at 37 °C in 96-well round-bottomed plates (for Act-mOVA H-2Kb-deficient cells with vaccinia virus–OVA priming and boosting) or in 96-well flat-bottomed plates for boosting with Listeria monocytogenes–OVA, in 200 μl Iscove's modified Dulbecco's medium plus OVA(257–264) (SIINFEKL) peptide) at a concentration of 1 μg/ml in the presence of GolgiPlug (BD Biosciences). Cells were stained with labeled phycoerythrin–Texas Red–conjugated anti-CD8 (MCD0817; Invitrogen), allophycocyanin–eFluor 780–conjugated anti-CD62L (MEL-14; eBioscience), Alexa Fluor 700–conjugated anti-CD44 (IM7; eBioscience), Pacific Blue–conjugated anti-CD45.1 (A20; BioLegend) and peridinin chlorophyll protein–cyanine 5.5–conjugated anti-CD45.2 (104; eBioscience) followed by fixation for 20 min at 4 °C. Fixed cells were subjected to intracellular cytokine with phycoerythrin-indotricarbocyanine–conjugated antibody to tumor necrosis factor (MP6-XT22; BD Pharmingen), allophycocyanin-conjugated anti-IFN-γ (XMG1.2; eBioscience) and phycoerythrin-conjugated anti-IL-2 (JES6-5H4; BioLegend). Samples were acquired on a LSR II (Becton Dickinson) and data were analyzed with FlowJo software.
Cytokine production in OT-II cells was assessed with the Fixation/Permeabilization Solution Kit with BD GolgiPlug according to the manufacturer's instructions (BD Pharmingen) after in vitro restimulation for 7 h at 37 °C in 96-well round-bottomed plates in 200 μl medium plus the I-Ab-binding peptide consisting of OVA amino acids 323–239 (10 μg/ml) before the addition of GolgiPlug for the final 5 h (BD Biosciences). Cells were stained for 20 min at 4 °C with labeled fluorescein isothiocyanate–conjugated anti-CD4 (GK1.5; eBioscience), anti-CD45.1, anti-CD44 and allophycocyanin—eFluor 780–conjugated anti-TCRβ (H57-597; eBioscience), followed by fixation with Cytofix/Cytoperm (BD Biosciences). Fixed cells were subjected to intracellular cytokine staining with anti-IFN-γ and anti-IL-2. Samples were acquired on a LSR II (Becton Dickinson) and data were analyzed with FlowJo software.
Tetramer staining and flow cytometry.
Cells were stained for 20 min at 25 °C with phycoerythrin-conjugated OVA(257–264)–H-2Kb tetramer (Beckman Coulter), followed by staining with anti-CD8, anti-CD62L, anti-CD44 and allophycocyanin-conjugated anti-CD127 (A7R34; eBioscience). Samples were acquired on an LSR II (Becton Dickinson) and data were analyzed with FlowJo software.
Granzyme B staining and flow cytometry.
Mice received 5 × 105 OT-I or OT-I Il2−/− (CD45.1+) cells 1 d before intravenous immunization at day 0 with 5 × 106 vaccinia virus–OVA. Blood samples were collected at days 3, 4 and 5 after immunization and cells were stained with anti-CD8 and anti-CD45.1, followed by fixation for 20 min at 4 °C with Cytofix/Cytoperm (BD Biosciences). Fixed cells were subjected to intracellular staining for 30 min at 4 °C in Perm/Wash buffer (BD Biosciences) with mouse immunoglobulin G1 isotype-matched control antibody (MG104; Invitrogen) or antibody to granzyme B (GRB04; Invitrogen). Samples were acquired on a LSR II (Becton Dickinson) and data were analyzed with FlowJo software.
In vivo cytotoxic assay.
Mice were injected with 5 × 103 OT-I or OT-I Il2−/− (CD45.1+) cells 1 d before intravenous immunization at day 0 with 1 × 106 PFU vaccinia virus–OVA. At day 5, 5 × 106 CD45.1+ splenocytes loaded with irrelevant peptide (adenovirus type 5 E1B, amino acids 192–200) and 5 × 106 CD45.1+ splenocytes loaded with specific peptide (OVA(257–264)) stained with 0.02 μM or 0.2 μM CFSE (carboxyfluorescein diacetate succinimidyl ester), respectively, were injected intravenously. Then, 15 h later, spleens were removed and samples acquired on an LSR II (Becton Dickinson) and subsequently analyzed with FlowJo software as reported10.
References
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272, 54–60 (1996).
van Stipdonk, M.J. et al. Dynamic programming of CD8+ T lymphocyte responses. Nat. Immunol. 4, 361–365 (2003).
van Stipdonk, M.J., Lemmens, E.E. & Schoenberger, S.P. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nat. Immunol. 2, 423–429 (2001).
Kaech, S.M. & Ahmed, R. Memory CD8+ T cell differentiation: initial antigen encounter triggers a developmental program in naive cells. Nat. Immunol. 2, 415–422 (2001).
Badovinac, V.P., Porter, B.B. & Harty, J.T. Programmed contraction of CD8+ T cells after infection. Nat. Immunol. 3, 619–626 (2002).
Masopust, D., Kaech, S.M., Wherry, E.J. & Ahmed, R. The role of programming in memory T-cell development. Curr. Opin. Immunol. 16, 217–225 (2004).
Sun, J.C. & Bevan, M.J. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science 300, 339–342 (2003).
Shedlock, D.J. & Shen, H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science 300, 337–339 (2003).
Bourgeois, C., Rocha, B. & Tanchot, C. A role for CD40 expression on CD8+ T cells in the generation of CD8+ T cell memory. Science 297, 2060–2063 (2002).
Janssen, E.M. et al. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 421, 852–856 (2003).
Janssen, E.M. et al. CD4+ T-cell help controls CD8+ T-cell memory via TRAIL-mediated activation-induced cell death. Nature 434, 88–93 (2005).
Badovinac, V.P., Messingham, K.A., Griffith, T.S. & Harty, J.T. TRAIL deficiency delays, but does not prevent, erosion in the quality of “helpless” memory CD8 T cells. J. Immunol. 177, 999–1006 (2006).
Bennett, S.R. et al. Help for cytotoxic-T-cell responses is mediated by CD40 signalling. Nature 393, 478–480 (1998).
Lanzavecchia, A. Immunology. Licence to kill. Nature 393, 413–414 (1998).
Ridge, J.P., Di Rosa, F. & Matzinger, P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature 393, 474–478 (1998).
Schoenberger, S.P., Toes, R.E., van der Voort, E.I., Offringa, R. & Melief, C.J. T-cell help for cytotoxic T lymphocytes is mediated by CD40–CD40L interactions. Nature 393, 480–483 (1998).
Horak, I., Lohler, J., Ma, A. & Smith, K.A. Interleukin-2 deficient mice: a new model to study autoimmunity and self-tolerance. Immunol. Rev. 148, 35–44 (1995).
Keene, J.A. & Forman, J. Helper activity is required for the in vivo generation of cytotoxic T lymphocytes. J. Exp. Med. 155, 768–782 (1982).
Mitchison, N.A. & O'Malley, C. Three-cell-type clusters of T cells with antigen-presenting cells best explain the epitope linkage and noncognate requirements of the in vivo cytolytic response. Eur. J. Immunol. 17, 1579–1583 (1987).
Williams, M.A., Tyznik, A.J. & Bevan, M.J. Interleukin-2 signals during priming are required for secondary expansion of CD8+ memory T cells. Nature 441, 890–893 (2006).
Kalia, V. et al. Prolonged interleukin-2Rα expression on virus-specific CD8+ T cells favors terminal-effector differentiation in vivo. Immunity 32, 91–103 (2010).
Pipkin, M.E. et al. Interleukin-2 and inflammation induce distinct transcriptional programs that promote the differentiation of effector cytolytic T cells. Immunity 32, 79–90 (2010).
Malek, T.R. & Bayer, A.L. Tolerance, not immunity, crucially depends on IL-2. Nat. Rev. Immunol. 4, 665–674 (2004).
Sadlack, B. et al. Generalized autoimmune disease in interleukin-2-deficient mice is triggered by an uncontrolled activation and proliferation of CD4+ T cells. Eur. J. Immunol. 25, 3053–3059 (1995).
Kurts, C. et al. Constitutive class I-restricted exogenous presentation of self antigens in vivo. J. Exp. Med. 184, 923–930 (1996).
Ehst, B.D., Ingulli, E. & Jenkins, M.K. Development of a novel transgenic mouse for the study of interactions between CD4 and CD8 T cells during graft rejection. Am. J. Transplant. 3, 1355–1362 (2003).
Nikolić-Zugić, J. & Bevan, M.J. Role of self-peptides in positively selecting the T-cell repertoire. Nature 344, 65–67 (1990).
Granucci, F., Feau, S., Angeli, V., Trottein, F. & Ricciardi-Castagnoli, P. Early IL-2 production by mouse dendritic cells is the result of microbial-induced priming. J. Immunol. 170, 5075–5081 (2003).
Badovinac, V.P., Haring, J.S. & Harty, J.T. Initial T cell receptor transgenic cell precursor frequency dictates critical aspects of the CD8+ T cell response to infection. Immunity 26, 827–841 (2007).
D'Souza, W.N. & Lefrancois, L. IL-2 is not required for the initiation of CD8 T cell cycling but sustains expansion. J. Immunol. 171, 5727–5735 (2003).
Kurts, C. et al. CD4+ T cell help impairs CD8+ T cell deletion induced by cross-presentation of self-antigens and favors autoimmunity. J. Exp. Med. 186, 2057–2062 (1997).
Wilson, E.B. & Livingstone, A.M. Cutting edge: CD4+ T cell-derived IL-2 is essential for help-dependent primary CD8+ T cell responses. J. Immunol. 181, 7445–7448 (2008).
Granucci, F. et al. A contribution of mouse dendritic cell-derived IL-2 for NK cell activation. J. Exp. Med. 200, 287–295 (2004).
Clarke, S.R. et al. Characterization of the ovalbumin-specific TCR transgenic line OT-I: MHC elements for positive and negative selection. Immunol. Cell Biol. 78, 110–117 (2000).
Castellino, F. et al. Chemokines enhance immunity by guiding naive CD8+ T cells to sites of CD4+ T cell-dendritic cell interaction. Nature 440, 890–895 (2006).
Kündig, T.M. et al. Immune responses in interleukin-2-deficient mice. Science 262, 1059–1061 (1993).
Macintyre, A.N. et al. Protein kinase B controls transcriptional programs that direct cytotoxic T cell fate but is dispensable for T cell metabolism. Immunity 34, 224–236 (2011).
Schorle, H., Holtschke, T., Hunig, T., Schimpl, A. & Horak, I. Development and function of T cells in mice rendered interleukin-2 deficient by gene targeting. Nature 352, 621–624 (1991).
Mach, N. et al. Differences in dendritic cells stimulated in vivo by tumors engineered to secrete granulocyte-macrophage colony-stimulating factor or Flt3-ligand. Cancer Res. 60, 3239–3246 (2000).
Henri, S. et al. The dendritic cell populations of mouse lymph nodes. J. Immunol. 167, 741–748 (2001).
Acknowledgements
We thank S. Salek-Ardakani (La Jolla Institute for Allergy and Immunology) for vaccinia virus–OVA; M.K. Jenkins (University of Minnesota Medical School) for Act-mOVA-transgenic mice; M. Rafii-El-Idrissi for demonstrating how to produce and purify the virus; F. Lambolez for the protocol for purifying T cells from the liver; S. Trifari and M. Pipkin for advice on granzyme B expression; and C. Kim and K. Van Gunst for technical advice and assistance. Supported by the US National Institutes of Health (R01 AI076972 and R01CA81261 to S.P.S.), the Leukemia and Lymphoma Society (1630-06 to S.P.S.) and the Kurz Family Foundation.
Author information
Authors and Affiliations
Contributions
S.F., R.A. and S.P.S. designed the experiments; S.F. and R.A. did the experiments with assistance from S.T.; and S.F. and S.P.S. analyzed the data and wrote the manuscript with contributions from R.A.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–2 (PDF 773 kb)
Rights and permissions
About this article
Cite this article
Feau, S., Arens, R., Togher, S. et al. Autocrine IL-2 is required for secondary population expansion of CD8+ memory T cells. Nat Immunol 12, 908–913 (2011). https://doi.org/10.1038/ni.2079
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2079
This article is cited by
-
Preclinical assessment of an anti-HTLV-1 heterologous DNA/MVA vaccine protocol expressing a multiepitope HBZ protein
Virology Journal (2023)
-
Blocking of oestrogen signals improves anti-tumour effect regardless of oestrogen receptor alpha expression in cancer cells
British Journal of Cancer (2023)
-
Resilient T-cell responses in patients with advanced cancers
International Journal of Hematology (2023)
-
Liver-resident memory T cells: life in lockdown
Seminars in Immunopathology (2022)
-
T cell signaling and Treg dysfunction correlate to disease kinetics in IL-2Rα-KO autoimmune mice
Scientific Reports (2020)