Abstract
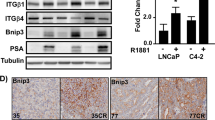
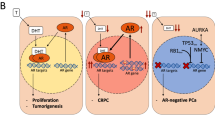
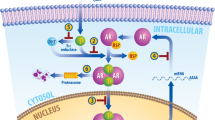
The transition from androgen-dependent to castration-resistant prostate cancer (CRPC) is a lethal event of uncertain molecular etiology. Comparing gene expression in isogenic androgen-dependent and CRPC xenografts, we found a reproducible increase in N-cadherin expression, which was also elevated in primary and metastatic tumors of individuals with CRPC. Ectopic expression of N-cadherin in nonmetastatic, androgen-dependent prostate cancer models caused castration resistance, invasion and metastasis. Monoclonal antibodies against the ectodomain of N-cadherin reduced proliferation, adhesion and invasion of prostate cancer cells in vitro. In vivo, these antibodies slowed the growth of multiple established CRPC xenografts, blocked local invasion and metastasis and, at higher doses, led to complete regression. N-cadherin–specific antibodies markedly delayed the time to emergence of castration resistance, markedly affected tumor histology and angiogenesis, and reduced both AKT serine-threonine kinase activity and serum interleukin-8 (IL-8) secretion. These data indicate that N-cadherin is a major cause of both prostate cancer metastasis and castration resistance. Therapeutic targeting of this factor with monoclonal antibodies may have considerable clinical benefit.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Chen, C.D. et al. Molecular determinants of resistance to antiandrogen therapy. Nat. Med. 10, 33–39 (2004).
Suzuki, H., Ueda, T., Ichikawa, T. & Ito, H. Androgen receptor involvement in the progression of prostate cancer. Endocr. Relat. Cancer 10, 209–216 (2003).
Mellado, B., Codony, J., Ribal, M.J., Visa, L. & Gascon, P. Molecular biology of androgen-independent prostate cancer: the role of the androgen receptor pathway. Clin. Transl. Oncol. 11, 5–10 (2009).
Harris, W.P., Mostaghel, E.A., Nelson, P.S. & Montgomery, B. Androgen deprivation therapy: progress in understanding mechanisms of resistance and optimizing androgen depletion. Nat. Clin. Pract. Urol. 6, 76–85 (2009).
Roudier, M.P. et al. Phenotypic heterogeneity of end-stage prostate carcinoma metastatic to bone. Hum. Pathol. 34, 646–653 (2003).
Shah, R.B. et al. Androgen-independent prostate cancer is a heterogeneous group of diseases: lessons from a rapid autopsy program. Cancer Res. 64, 9209–9216 (2004).
Lassi, K. & Dawson, N.A. Emerging therapies in castrate-resistant prostate cancer. Curr. Opin. Oncol. 21, 260–265 (2009).
Attard, G., Reid, A.H., Olmos, D. & de Bono, J.S. Antitumor activity with CYP17 blockade indicates that castration-resistant prostate cancer frequently remains hormone driven. Cancer Res. 69, 4937–4940 (2009).
Rizzi, F. & Bettuzzi, S. Targeting clusterin in prostate cancer. J. Physiol. Pharmacol. 59 Suppl 9, 265–274 (2008).
Gleave, M., Miyake, H. & Chi, K. Beyond simple castration: targeting the molecular basis of treatment resistance in advanced prostate cancer. Cancer Chemother. Pharmacol. 56 Suppl 1, 47–57 (2005).
Sharifi, N., Kawasaki, B.T., Hurt, E.M. & Farrar, W.L. Stem cells in prostate cancer: resolving the castrate-resistant conundrum and implications for hormonal therapy. Cancer Biol. Ther. 5, 901–906 (2006).
Isaacs, J.T. The biology of hormone refractory prostate cancer. Why does it develop? Urol. Clin. North Am. 26, 263–273 (1999).
Gu, Z. et al. Reg IV: a promising marker of hormone refractory metastatic prostate cancer. Clin. Cancer Res. 11, 2237–2243 (2005).
Tso, C.L. et al. Androgen deprivation induces selective outgrowth of aggressive hormone-refractory prostate cancer clones expressing distinct cellular and molecular properties not present in parental androgen-dependent cancer cells. Cancer J. 6, 220–233 (2000).
Tran, N.L., Adams, D.G., Vaillancourt, R.R. & Heimark, R.L. Signal transduction from N-cadherin increases Bcl-2. Regulation of the phosphatidylinositol 3-kinase/Akt pathway by homophilic adhesion and actin cytoskeletal organization. J. Biol. Chem. 277, 32905–32914 (2002).
Araki, S. et al. Interleukin-8 is a molecular determinant of androgen independence and progression in prostate cancer. Cancer Res. 67, 6854–6862 (2007).
Domingo-Domenech, J. et al. Interleukin 6, a nuclear factor-κB target, predicts resistance to docetaxel in hormone-independent prostate cancer and nuclear factor-κB inhibition by PS-1145 enhances docetaxel antitumor activity. Clin. Cancer Res. 12, 5578–5586 (2006).
Tomita, K. et al. Cadherin switching in human prostate cancer progression. Cancer Res. 60, 3650–3654 (2000).
Jaggi, M. et al. N-cadherin switching occurs in high Gleason grade prostate cancer. Prostate 66, 193–199 (2006).
Gravdal, K., Halvorsen, O.J., Haukaas, S.A. & Akslen, L.A. A switch from E-cadherin to N-cadherin expression indicates epithelial to mesenchymal transition and is of strong and independent importance for the progress of prostate cancer. Clin. Cancer Res. 13, 7003–7011 (2007).
Mani, S.A. et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133, 704–715 (2008).
Mason, M.J., Fan, G., Plath, K., Zhou, Q. & Horvath, S. Signed weighted gene co-expression network analysis of transcriptional regulation in murine embryonic stem cells. BMC Genomics 10, 327 (2009).
Majumder, P.K. & Sellers, W.R. Akt-regulated pathways in prostate cancer. Oncogene 24, 7465–7474 (2005).
Kim, J.B. et al. N-Cadherin extracellular repeat 4 mediates epithelial to mesenchymal transition and increased motility. J. Cell Biol. 151, 1193–1206 (2000).
Li, J. et al. Cardiac-specific loss of N-cadherin leads to alteration in connexins with conduction slowing and arrhythmogenesis. Circ. Res. 97, 474–481 (2005).
Klein, K.A. et al. Progression of metastatic human prostate cancer to androgen independence in immunodeficient SCID mice. Nat. Med. 3, 402–408 (1997).
Hara, T., Miyazaki, H., Lee, A., Tran, C.P. & Reiter, R.E. Androgen receptor and invasion in prostate cancer. Cancer Res. 68, 1128–1135 (2008).
Gu, Z. et al. Prostate stem cell antigen (PSCA) expression increases with high Gleason score, advanced stage and bone metastasis in prostate cancer. Oncogene 19, 1288–1296 (2000).
Acknowledgements
This work was supported in parts by the US National Cancer Institute Prostate Cancer SPORE at the University of California–Los Angeles (P50CA092131-09 to R.E.R.), US Department of Defense Prostate Cancer Research grants (W81XWH-06-1-0324 to Z.A.W., W81XWH-09-1-0630 to R.E.R. and M.B.R., PC061456 to J.H.), Takeda Pharmaceuticals, the Jean Perkins Foundation and the American Cancer Society (RSG-07-092-01-TBE to J.H.). We also thank S. and L. Resnick, the Prostate Cancer Foundation and the Luskin Foundation for generous support and J. Said and N. Doan for immunohistochemical assessments. LNCaP-CL1 cells were provided by C.L. Tso (University of California–Los Angeles). Plasmid pΔVPR was provided by I. Chen (University of California–Los Angeles).
Author information
Authors and Affiliations
Contributions
H.T. and E.K. designed and conducted in vitro and in vivo studies. C.P.T. generated stable N-cadherin–knockdown reagents and prepared the manuscript. H.M. made the N-cadherin–overexpressing cell lines. J.Y. and R.W. performed gene and protein expression analyses. T.S. contributed to the in vivo N-cadherin–knockdown and antibody studies. F.L. and M.G. conducted immunohistochemical evaluation of prostate cancer specimens. J.H. contributed to immunohistochemical analyses of in vivo studies. R.L.V. provided clinical materials for the initial N-cadherin screening in metastases. J.A. and M.B.R. provided data on AKT activity. S.H. performed gene expression analysis for stem cell markers. Z.A.W. generated the monoclonal antibodies. R.E.R. conceived of the study and supervised the project. All authors discussed the results and commented on the manuscript at all stages.
Corresponding author
Ethics declarations
Competing interests
R.E.R. has an equity interest in EMTx Therapeutics, which has an option to license and develop N-cadherin–specific antibodies for cancer therapy.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 1282 kb)
Rights and permissions
About this article
Cite this article
Tanaka, H., Kono, E., Tran, C. et al. Monoclonal antibody targeting of N-cadherin inhibits prostate cancer growth, metastasis and castration resistance. Nat Med 16, 1414–1420 (2010). https://doi.org/10.1038/nm.2236
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2236
This article is cited by
-
MAP4K4 promotes ovarian cancer metastasis through diminishing ADAM10-dependent N-cadherin cleavage
Oncogene (2023)
-
Nanosecond pulsed electric field suppresses growth and reduces multi-drug resistance effect in pancreatic cancer
Scientific Reports (2023)
-
Mechanical stress shapes the cancer cell response to neddylation inhibition
Journal of Experimental & Clinical Cancer Research (2022)
-
Mesoporous nanocarriers with cyclic peptide gatekeeper containing N-cadherin binding sequence for stimulus-responsive drug release
Chemical Papers (2022)
-
Cellular rewiring in lethal prostate cancer: the architect of drug resistance
Nature Reviews Urology (2020)