Abstract
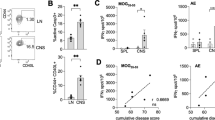
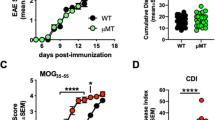
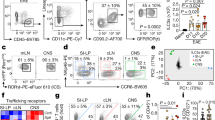
Chronic progression of two T cell–mediated central nervous system (CNS) demyelinating models of multiple sclerosis, relapsing EAE (R-EAE) and Theiler's murine encephalomyelitis virus–induced demyelinating disease (TMEV-IDD) is dependent on the activation of T cells to endogenous myelin epitopes (epitope spreading). Using transfer of carboxyfluorescein succinyl ester (CFSE)-labeled T-cell receptor (TCR)-transgenic T cells and mixed bone marrow chimeras, we show that activation of naive proteolipid protein (PLP)139–151-specific T cells in SJL mice undergoing PLP178–191-induced R-EAE or TMEV-IDD occurs directly in the CNS and not in the cervical lymph nodes or other peripheral lymphoid organs. Examination of the antigen-presentation capacity of antigen-presenting cell (APC) populations purified from the CNS of mice with PLP178–191-induced R-EAE shows that only F4/80−CD11c+CD45hi dendritic cells (DCs) efficiently present endogenous antigen to activate naive PLP139–151-specific T cells in vitro. In contrast, DCs as well as F4/80+CD45hi macrophages and F4/80+CD45lo microglia activate a PLP139–151-specific helper T cell line. The data suggest that naive T cells enter the inflamed CNS and are activated by local APCs, possibly DCs, to initiate epitope spreading.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vanderlugt, C.L. & Miller, S.D. Epitope spreading in immune-mediated diseases: implications for immunotherapy. Nat. Rev. Immunol. 2, 85–95 (2002).
Tuohy, V.K., Yu, M., Yin, L., Kawczak, J.A. & Kinkel, R.P. Spontaneous regression of primary autoreactivity during chronic progression of experimental autoimmune encephalomyelitis and multiple sclerosis. J. Exp. Med. 189, 1033–1042 (1999).
Lehmann, P.V., Forsthuber, T., Miller, A. & Sercarz, E.E. Spreading of T-cell autoimmunity to cryptic determinants of an autoantigen. Nature 358, 155–157 (1992).
McRae, B.L., Vanderlugt, C.L., Dal Canto, M.C. & Miller, S.D. Functional evidence for epitope spreading in the relapsing pathology of experimental autoimmune encephalomyelitis. J. Exp. Med. 182, 75–85 (1995).
Vanderlugt, C.L. et al. Pathologic role and temporal appearance of newly emerging autoepitopes in relapsing experimental autoimmune encephalomyelitis. J. Immunol. 164, 670–678 (2000).
Yu, M., Johnson, J.M. & Tuohy, V.K. A predictable sequential determinant spreading cascade invariably accompanies progression of experimental autoimmune encephalomyelitis: A basis for peptide-specific therapy after onset of clinical disease. J. Exp. Med. 183, 1777–1788 (1996).
Miller, S.D. et al. Persistent infection with Theiler's virus leads to CNS autoimmunity via epitope spreading. Nat. Med. 3, 1133–1136 (1997).
Katz-Levy, Y. et al. Temporal development of autoreactive Th1 responses and endogenous antigen presentation of self myelin epitopes by CNS-resident APCs in Theiler's virus-infected mice. J. Immunol. 165, 5304–5314 (2000).
Neville, K.L., Padilla, J. & Miller, S.D. Myelin-specific tolerance attenuates the progression of a virus-induced demyelinating disease: implications for the treatment of MS. J. Neuroimmunol. 123, 18–29 (2002).
Yamada, S., DePasquale, M., Patlak, C.S. & Cserr, H.F. Albumin outflow into deep cervical lymph from different regions of rabbit brain. Am. J. Physiol. 261, H1197–H1204 (1991).
Hochwald, G.M., Van, D.A., Robinson, M.E. & Thorbecke, G.J. Immune response in draining lymph nodes and spleen after intraventricular injection of antigen. Int. J. Neurosci. 39, 299–306 (1988).
Ling, C., Sandor, M. & Fabry, Z. In situ processing and distribution of intracerebrally injected OVA in the CNS. J. Neuroimmunol. 141, 90–98 (2003).
Karman, J., Ling, C., Sandor, M. & Fabry, Z. Initiation of immune responses in brain is promoted by local dendritic cells. J. Immunol. 173, 2353–2361 (2004).
de Vos, A.F. et al. Transfer of central nervous system autoantigens and presentation in secondary lymphoid organs. J. Immunol. 169, 5415–5423 (2002).
Hickey, W.F. & Kimura, H. Perivascular microglial cells of the CNS are bone marrow-derived and present antigen in vivo. Science 239, 290–292 (1988).
Mack, C.L., Neville, K.L. & Miller, S.D. Microglia are activated to become competent antigen presenting and effector cells in the inflammatory environment of the Theiler's virus model of multiple sclerosis. J. Neuroimmunol. 144, 68–79 (2003).
Serafini, B., Columba-Cabezas, S., Di, R.F. & Aloisi, F. Intracerebral recruitment and maturation of dendritic cells in the onset and progression of experimental autoimmune encephalomyelitis. Am. J. Pathol. 157, 1991–2002 (2000).
Fischer, H.G. & Reichmann, G. Brain dendritic cells and macrophages/microglia in central nervous system inflammation. J. Immunol. 166, 2717–2726 (2001).
Kearney, E.R., Pape, K.A., Loh, D.Y. & Jenkins, M.K. Visualization of peptide-specific T cell immunity and peripheral tolerance induction in vivo. Immunity 1, 327–339 (1994).
Oehmichen, M., Gruninger, H., Wietholter, H. & Gencic, M. Lymphatic efflux of intracerebrally injected cells. Acta Neuropathol. (Berl) 45, 61–65 (1979).
Ngo, V.N. et al. Lymphotoxin alpha/beta and tumor necrosis factor are required for stromal cell expression of homing chemokines in B and T cell areas of the spleen. J. Exp. Med. 189, 403–412 (1999).
Muldoon, L.L. et al. Trafficking of superparamagnetic iron oxide particles (Combidex) from brain to lymph nodes in the rat. Neuropathol. Appl. Neurobiol. 30, 70–79 (2004).
Suter, T. et al. Dendritic cells and differential usage of the MHC class II transactivator promoters in the central nervous system in experimental autoimmune encephalitis. Eur. J. Immunol. 30, 794–802 (2000).
Pashenkov, M. et al. Two subsets of dendritic cells are present in human cerebrospinal fluid. Brain 124, 480–492 (2001).
Plumb, J., Armstrong, M.A., Duddy, M., Mirakhur, M. & McQuaid, S. CD83-positive dendritic cells are present in occasional perivascular cuffs in multiple sclerosis lesions. Mult. Scler. 9, 142–147 (2003).
Kivisakk, P. et al. Expression of CCR7 in multiple sclerosis: implications for CNS immunity. Ann. Neurol. 55, 627–638 (2004).
Krakowski, M.L. & Owens, T. Naive T lymphocytes traffic to inflamed central nervous system, but require antigen recognition for activation. Eur. J. Immunol. 30, 1002–1009 (2000).
Greter, M. et al. Dendritic cells permit immune invasion of the CNS during experimental autoimmune encephalomyelitis. Nat. Med. 11, 328–334 (2005).
Waldner, H., Whitters, M.J., Sobel, R.A., Collins, M. & Kuchroo, V.K. Fulminant spontaneous autoimmunity of the central nervous system in mice transgenic for the myelin proteolipid protein-specific T cell receptor. Proc. Natl. Acad. Sci. USA 97, 3412–3417 (2000).
Tompkins, S.M. et al. De novo central nervous system processing of myelin antigen is required for the initiation of experimental autoimmune encephalomyelitis. J. Immunol. 168, 4173–4183 (2002).
Acknowledgements
We thank J. Marvin at the University of Chicago for FACS-sorting expertise and A. Kohm for critical reading of the manuscript. The authors acknowledge the contribution of discussion with colleagues at the Myelin Repair Foundation. Supported in part by grants from the US National Institutes of Health (NS-30871 and NS-23349) and the Myelin Repair Foundation. E.J.M. was supported by National Institutes of Health Training Grant AI-07476 and S.L.B. by National Multiple Sclerosis Society Postdoctoral Fellowship Grant FG 1563 A-1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
PLP139–151 reactivity in PLP178–191-primed mice. (PDF 113 kb)
Supplementary Fig. 2
FACS purification of CNS antigen presenting cell populations. (PDF 124 kb)
Supplementary Fig. 3
CD11c+ DCs are found in the same areas as CD4+ T cells in the CNS of mice with both R-EAE and TMEV-IDD. (PDF 161 kb)
Rights and permissions
About this article
Cite this article
McMahon, E., Bailey, S., Castenada, C. et al. Epitope spreading initiates in the CNS in two mouse models of multiple sclerosis. Nat Med 11, 335–339 (2005). https://doi.org/10.1038/nm1202
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1202
This article is cited by
-
Tissue-resident immune cells in the pathogenesis of multiple sclerosis
Inflammation Research (2023)
-
Immunosuppressive biomaterial-based therapeutic vaccine to treat multiple sclerosis via re-establishing immune tolerance
Nature Communications (2022)
-
Interleukin-31 and soluble CD40L: new candidate serum biomarkers that predict therapeutic response in multiple sclerosis
Neurological Sciences (2022)
-
Homotaurine limits the spreading of T cell autoreactivity within the CNS and ameliorates disease in a model of multiple sclerosis
Scientific Reports (2021)
-
Co-delivery of IOX1 and doxorubicin for antibody-independent cancer chemo-immunotherapy
Nature Communications (2021)