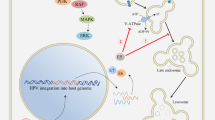
Abstract
Tumour-associated viruses produce antigens that, on the face of it, are ideal targets for immunotherapy. Unfortunately, these viruses are experts at avoiding or subverting the host immune response. Cervical-cancer-associated human papillomavirus (HPV) has a battery of immune-evasion mechanisms at its disposal that could confound attempts at HPV-directed immunotherapy. Other virally associated human cancers might prove similarly refractive to immuno-intervention unless we learn how to circumvent their strategies for immune evasion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zhou, J., Liu, W. J., Peng, S. W., Sun, X. Y. & Frazer, I. Papillomavirus capsid protein expression level depends on the match between codon usage and tRNA availability. J. Virol. 73, 4972–4982 (1999).
Schwartz, S. Regulation of human papillomavirus late gene expression. Ups. J. Med. Sci. 105, 171–192 (2000).
Nees, M. et al. Papillomavirus type 16 oncogenes downregulate expression of interferon-responsive genes and upregulate proliferation-associated and NF-κB-responsive genes in cervical keratinocytes. J. Virol. 75, 4283–4296 (2001).
Barnard, P., Payne, E. & McMillan, N. A. The human papillomavirus E7 protein is able to inhibit the antiviral and anti-growth functions of interferon-α. Virology 277, 411–419 (2000).
Park, J. S. et al. Inactivation of interferon regulatory factor-1 tumor suppressor protein by HPV E7 oncoprotein. Implication for the E7-mediated immune evasion mechanism in cervical carcinogenesis. J. Biol. Chem. 275, 6764–6769 (2000).
Arany, I., Goel, A. & Tyring, S. K. Interferon response depends on viral transcription in human papillomavirus containing lesions. Anticancer Res. 15, 2865–2870 (1995).
Rescigno, M. et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nature Immunol. 2, 361–367 (2001).
Li, M. et al. Cell-associated ovalbumin is cross-presented much more efficiently than soluble ovalbumin in vivo. J. Immunol. 166, 6099–6103 (2001).
Wolfers, J. et al. Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nature Med. 7, 297–303 (2001).
Frazer, I. H. et al. Systemic administration of bacterial products induces host protective specific immunity to an epithelial tumour antigen. J. Immunol. 167, 6180–6187 (2001).
Doan, T. et al. Human papillomavirus type 16 E7 oncoprotein expressed in peripheral epithelium tolerizes E7-directed cytotoxic T-lymphocyte precursors restricted through human (and mouse) major histocompatibility complex class I alleles. J. Virol. 73, 6166–6170 (1999).
Giannini, S. L. et al. Cytokine expression in squamous intraepithelial lesions of the uterine cervix: implications for the generation of local immunosuppression. Clin. Exp. Immunol. 113, 183–189 (1998).
Geissmann, F. et al. TGF-β1 prevents the noncognate maturation of human dendritic Langerhans cells. J. Immunol. 162, 4567–4575 (1999).
Hemmi, H. et al. Skin antigens in the steady state are trafficked to regional lymph nodes by transforming growth factor-β1-dependent cells. Int. Immunol. 13, 695–704 (2001).
Hoffmann, T. K., Meidenbauer, N., Dworacki, G., Kanaya, H. & Whiteside, T. L. Generation of tumor-specific T-lymphocytes by cross-priming with human dendritic cells ingesting apoptotic tumor cells. Cancer Res. 60, 3542–3549 (2000).
Albert, M. L., Jegathesan, M. & Darnell, R. B. Dendritic cell maturation is required for the cross-tolerization of CD8+ T cells. Nature Immunol. 2, 1010–1017 (2001).
Suri-Payer, E., Amar, A. Z., Thornton, A. M. & Shevach, E. M. CD4+CD25+ T cells inhibit both the induction and effector function of autoreactive T cells and represent a unique lineage of immunoregulatory cells. J. Immunol. 160, 1212–1218 (1998).
Grohmann, U. et al. CD40 ligation ablates the tolerogenic potential of lymphoid dendritic cells. J. Immunol. 166, 277–283 (2001).
Dhodapkar, M. V., Steinman, R. M., Krasovsky, J., Munz, C. & Bhardwaj, N. Antigen-specific inhibition of effector T cell function in humans after injection of immature dendritic cells. J. Exp. Med. 193, 233–238 (2001).
Roncarolo, M. G., Levings, M. K. & Traversari, C. Differentiation of T regulatory cells by immature dendritic cells. J. Exp. Med. 193, F5–F9 (2001).
Corinti, S., Albanesi, C., la Sala, A., Pastore, S. & Girolomoni, G. Regulatory activity of autocrine IL-10 on dendritic cell functions. J. Immunol. 166, 4312–4318 (2001).
Ressing, M. E. et al. Human CTL epitopes encoded by human papillomavirus type 16 E6 and E7 identified through in vitro and in vivo immunogenicity studies of HLA-A*0201-binding peptides. J. Immunol. 154, 5934–5943 (1995).
Tindle, R. W. et al. Nonspecific down-regulation of CD8+ T-cell responses in mice expressing human papillomavirus type 16 E7 oncoprotein from the keratin-14 promoter. J. Virol. 75, 5985–5997 (2001).
Lambrecht, B. N., Pauwels, R. A. & Fazekas De St Groth, B. Induction of rapid T cell activation, division, and recirculation by intratracheal injection of dendritic cells in a TCR transgenic model. J. Immunol. 164, 2937–2946 (2000).
Harshyne, L. A., Watkins, S. C., Gambotto, A. & Barratt-Boyes, S. M. Dendritic cells acquire antigens from live cells for cross-presentation to CTL. J. Immunol. 166, 3717–3723 (2001).
Natale, C., Giannini, T., Lucchese, A. & Kanduc, D. Computer-assisted analysis of molecular mimicry between human papillomavirus 16 E7 oncoprotein and human protein sequences. Immunol. Cell Biol. 78, 580–585 (2000).
Lee, S. J. et al. Both E6 and E7 oncoproteins of human papillomavirus 16 inhibit IL-18- induced IFN-γ production in human peripheral blood mononuclear and NK cells. J. Immunol. 167, 497–504 (2001).
Le Buanec, H. et al. Induction of cellular immunosuppression by the human papillomavirus type 16 E7 oncogenic protein. Biomed. Pharmacother. 53, 323–328 (1999).
Cho, Y.-S. et al. Down modulation of IL-18 expression by human papillomavirus type 16 E6 oncogene via binding to IL-18. FEBS Lett. 501, 139–145 (2001).
Straight, S. W., Herman, B. & McCance, D. J. The E5 oncoprotein of human papillomavirus type 16 inhibits the acidification of endosomes in human keratinocytes. J. Virol. 69, 3185–3192 (1995).
Gabrilovich, D. I., Velders, M. P., Sotomayor, E. M. & Kast, W. M. Mechanism of immune dysfunction in cancer mediated by immature Gr-1+ myeloid cells. J. Immunol. 166, 5398–5406 (2001).
Sheu, B. C. et al. A novel role of metalloproteinase in cancer-mediated immunosuppression. Cancer Res. 61, 237–242 (2001).
Brady, C. S. et al. Multiple mechanisms underlie HLA dysregulation in cervical cancer. Tissue Antigens 55, 401–411 (2000).
Evans, M. et al. Antigen processing defects in cervical carcinomas limit the presentation of a CTL epitope from human papillomavirus 16 E6. J. Immunol. 167, 5420–5428 (2001).
Wang, S. S. et al. Human leucocyte antigen class I and II alleles and risk of cervical neoplasia: results from a population-based study in Costa Rica. J. Infect. Dis. 184, 1310–1314 (2001).
Krul, E. J. T. et al. HLA susceptibility to cervical neoplasia. Hum. Immunol. 60, 337–342 (1999).
Sasaki, S., Amara, R. R., Oran. A. E., Smith, J. M. & Robinson, H. L. Apoptosis-mediated enhancement of DNA-raised immune responses by mutant caspases. Nature Biotechnol. 19, 543–547 (2001).
Restifo, N. P. Building better vaccines: how apoptotic cell death can induce inflammation and activate innate and adaptive immunity. Curr. Opin. Immunol. 12, 597–603 (2000).
Biragyn, A. & Kwak, L. W. Designer cancer vaccines are still in fashion. Nature Med. 6, 966–968 (2000).
Fabre, J. W. The allogeneic response and tumor immunity. Nature Med. 7, 649–652 (2001).
Coussens, L. M., Tinkle, C. L., Hanahan, D. & Werb, Z. MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 103, 481–490 (2000).
Rosenberg, S. A. Progress in human tumour immunology and immunotherapy. Nature 411, 380–384 (2001).
Muller, A., Schmitt, L., Raftery, M. & Schonrich, G. Paralysis of B7 co-stimulation through the effect of viral IL-10 on T cells as a mechanism of local tolerance induction. Eur. J. Immunol. 28, 3488–3498 (1998).
Huang, H., Pan, X. & Zhou, J. BHRF1 antisense oligonucleotide inhibits anti-apoptosis of nasopharyngeal carcinoma cells. Int. J. Mol. Med. 4, 649–653 (1999).
Sette, A. D. et al. Overcoming T cell tolerance to the hepatitis B virus surface antigen in hepatitis B virus-transgenic mice. J. Immunol. 166, 1389–1397 (2001).
Anderson, C. C. et al. Testing time-, ignorance-, and danger-based models of tolerance. J. Immunol. 166, 3663–3671 (2001).
Fernando, G. J., Stewart, T. J., Tindle, R. W. & Frazer, I. H. Th2-type CD4+ cells neither enhance nor suppress antitumor CTL activity in a mouse tumor model. J. Immunol. 161, 2421–2427 (1998).
Acknowledgements
The author thanks R. Thomas for helpful discussion.
Author information
Authors and Affiliations
Related links
Glossary
- ADJUVANT
-
Any substance that nonspecifically enhances the immune response to antigen.
- ALLOGENEIC
-
Inter-individual genetic variation at the MHC locus. In the semi-allogeneic situation reported in this article, MHC antigens are shared by donor and recipient, but in addition the donor has some MHC antigens that the recipient does not
- ANERGY
-
A state in which T cells cannot respond to antigen.
- ANTIGEN-PRESENTING CELLS (APCs)
-
Cells that process antigen and present antigen fragments to other cells of the immune system (cross-presentation) to initiate an immune response. Dendritic cells are the most potent APCs.
- CD4 HELP
-
The secretion of cytokines, particularly interleukin-2, that are necessary for the expression of effector function by other cells in the immune system, especially CD8+ T cells.
- CD40 LIGAND
-
A molecule on CD4+ T cells that, when it binds CD40 on dendritic cells, stimulates them to mature.
- CD8+ T CELL
-
T cell bearing the CD8+ cell-surface glycoprotein, which recognizes MHC class I molecules on target cells. CD8+ T cells are usually cytotoxic T cells.
- CONDYLOMA
-
Papillomavirus-associated soft wart on genital mucosal epithelium.
- DECOY CYTOKINE
-
A molecule encoded by the genome of an invading organism that mimics the effect of a host cytokine and manipulates the immune response to the invader's advantage.
- EARLY PROTEINS
-
Gene products translated from the first mRNAs that are produced from the viral genome, before viral replication gets underway.
- GENITAL HYPERPLASIA
-
Dysregulated proliferation of keratinocytes of genital epithelium.
- LANGERHANS CELLS
-
A subset of antigen-presenting cells in the epithelium.
- LATE PROTEINS.
-
After viral DNA replication, 'late' viral genes become available for transcription. Their mRNAs encode structural proteins (L1 and L2 in HPV), which form the capsids of new virions.
- MAJOR HISTOCOMPATIBILITY COMPLEX (MHC)
-
A genetic region encoding proteins that are involved in antigen presentation to T cells. MHC class I molecules bound to peptide are recognized by the T-cell receptors of CD8+ T cells.
- MATURATION OF DENDRITIC CELLS
-
A process that is instigated by inflammatory cytokines, in which dendritic cells acquire co-stimulatory molecules that allow them to interact with and transmit activating signal(s) to specific T cells.
- NATURAL KILLER (NK) CELLS
-
Large granular lymphocytes that do not bear a T-cell receptor, but can recognize and destroy certain tumour cells and virally infected cells in an MHC-independent manner.
- REDUNDANCY IN THE GENETIC CODE
-
Several nucleotide triplets can encode the same amino acid and, correspondingly, several tRNAs are observed for that amino acid. Differences in abundance of different tRNAs are observed between species and sometimes between tissues in the same species.
- SYNGENEIC
-
Genetically identical, for example, a fully inbred strain of mouse.
- TRANSPORTER ASSOCIATED WITH ANTIGEN PROCESSING (TAP)
-
The peptide products of protein degradation in cytoplasm are transported by TAP molecules to the endoplasmic reticulum where they are loaded onto MHC molecules.
Rights and permissions
About this article
Cite this article
Tindle, R. Immune evasion in human papillomavirus-associated cervical cancer. Nat Rev Cancer 2, 59–64 (2002). https://doi.org/10.1038/nrc700
Issue Date:
DOI: https://doi.org/10.1038/nrc700
This article is cited by
-
Rescue of NLRC5 expression restores antigen processing machinery in head and neck cancer cells lacking functional STAT1 and p53
Cancer Immunology, Immunotherapy (2024)
-
Association between common vaginal and HPV infections and results of cytology test in the Zhoupu District, Shanghai City, China, from 2014 to 2019
Virology Journal (2022)
-
Human papillomavirus and cervical cancer: an insight highlighting pathogenesis and targeting strategies
VirusDisease (2022)
-
An efficient method to identify virus-specific TCRs for TCR-T cell immunotherapy against virus-associated malignancies
BMC Immunology (2021)
-
Baseline immunity and impact of chemotherapy on immune microenvironment in cervical cancer
British Journal of Cancer (2021)