Abstract
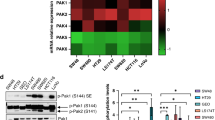
KRAS mutation is a predictive biomarker for resistance to cetuximab (Erbitux) in metastatic colorectal cancer (mCRC). This study sought to determine if KRAS mutant CRC lines could be sensitized to cetuximab using dasatinib (BMS-354825, Sprycel), a potent, orally bioavailable inhibitor of several tyrosine kinases, including the Src family kinases (SFKs). We analyzed 16 CRC lines for: (1) KRAS mutation status, (2) dependence on mutant KRAS signaling and (3) expression level of epidermal growth factor receptor (EGFR) and SFKs. From these analyses, we selected three KRAS mutant (LS180, LoVo and HCT116) cell lines and two KRAS wild-type cell lines (SW48 and CaCo2). In vitro, using poly-D-lysine/laminin plates, KRAS mutant cell lines were resistant to cetuximab, whereas KRAS wild-type lines showed sensitivity to cetuximab. Treatment with cetuximab and dasatinib showed a greater antiproliferative effect on KRAS mutant lines when compared with either agent alone in vitro and in vivo. To investigate potential mechanisms for this antiproliferative response in the combinatorial therapy, we performed Human Phospho-Kinase Antibody Array analysis, measuring the relative phosphorylation levels of 39 intracellular proteins in untreated, cetuximab, dasatinib or the combinatorial treatment in the KRAS mutant lines LS180, LoVo and HCT116 cells. The results of this experiment showed a decrease in a broad spectrum of kinases centered on the β-catenin pathway, the mitogen-activated protein kinase (MAPK) pathway, AKT/mammalian target of rapamycin (mTOR) pathway and the family of signal transducers and activators of transcription (STATs) when compared with the untreated control or monotherapy treatments. Next, we analyzed tumor growth with cetuximab, dasatinib or their combination in vivo. KRAS mutant xenografts showed resistance to cetuximab therapy, whereas KRAS wild type demonstrated an antitumor response when treated with cetuximab. KRAS mutant tumors exhibited minimal response to dasatinib monotherapy. However, as in vitro, KRAS mutant lines exhibited a response to the combination of cetuximab and dasatinib. Combinatorial treatment of KRAS mutant xenografts resulted in decreased cell proliferation, as measured by Ki67, and higher rates of apoptosis, as measured by TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling). The data presented in this study indicate that dasatinib can sensitize KRAS mutant CRC tumors to cetuximab and may do so by altering the activity of several key signaling pathways. Furthermore, these results suggest that signaling via EGFR and SFKs may be necessary for cell proliferation and survival of KRAS mutant CRC tumors. These data strengthen the rationale for clinical trials combining cetuximab and dasatinib in the KRAS mutant CRC genetic setting.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- ABL:
-
v-abl Abelson murin leukemia viral oncogene homolog 1
- BCR:
-
breakpoint cluster region
- CRC:
-
colorectal cancer
- EGFR:
-
epidermal growth factor receptor
- eNOS:
-
endothelial nitric oxide synthase
- FAK:
-
focal adhesion kinase-1
- FDA:
-
Food and Drug Administration
- GSK α/β:
-
glycogen synthase kinase α/β
- HNSCC:
-
head and neck squamous cell carcinoma
- IgG:
-
immunoglobulin G
- IHC:
-
immunohistochemistry
- MAPK:
-
mitogen-activated protein kinase
- mCRC:
-
metastatic colorectal cancer
- MEK:
-
MAPK kinase
- MSK:
-
mitogen and stress-activated protein kinase
- mTOR:
-
mammalian target of rapamycin
- NSCLC:
-
non-small cell lung cancer
- PDL:
-
poly-D-lysine
- PDGFR:
-
platelet-derived growth factor receptor
- PI3K:
-
phosphatidylinositol 3-kinase
- PKC:
-
protein kinase C
- PLCγ:
-
phospholipase C-γ
- RSK:
-
ribosomal s6 kinase
- RTK:
-
receptor tyrosine kinase
- SFK:
-
Src-family kinase
- STAT:
-
signal transducer and activator of transcription
- TUNEL:
-
terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling
References
Abram CL, Courtneidge SA . (2000). Src family tyrosine kinases and growth factor signaling. Exp Cell Res 254: 1–13.
Aligayer H, Boyd DD, Heiss MM, Abdalla EK, Curley SA, Gallick GE . (2002). Activation of Src kinase in primary colorectal carcinoma: an indicator of poor clinical prognosis. Cancer 94: 344–351.
Allegra CJ, Jessup JM, Somerfield MR, Hamilton SR, Hammond EH, Hayes DF et al. (2009). American Society of Clinical Oncology provisional clinical opinion: testing for KRAS gene mutations in patients with metastatic colorectal carcinoma to predict response to anti-epidermal growth factor receptor monoclonal antibody therapy. J Clin Oncol 27: 2091–2096.
Bardelli A, Siena S . (2010). Molecular mechanisms of resistance to cetuximab and panitumumab in colorectal cancer. J Clin Oncol 28: 1254–1261.
Belsches AP, Haskell MD, Parsons SJ . (1997). Role of c-Src tyrosine kinase in EGF-induced mitogenesis. Front Biosci 2: d501–d518.
Belsches-Jablonski AP, Biscardi JS, Peavy DR, Tice DA, Romney DA, Parsons SJ . (2001). Src family kinases and HER2 interactions in human breast cancer cell growth and survival. Oncogene 20: 1465–1475.
Benvenuti S, Sartore-Bianchi A, Di Nicolantonio F, Zanon C, Moroni M, Veronese S et al. (2007). Oncogenic activation of the RAS/RAF signaling pathway impairs the response of metastatic colorectal cancers to anti-epidermal growth factor receptor antibody therapies. Cancer Res 67: 2643–2648.
Biscardi JS, Ishizawar RC, Silva CM, Parsons SJ . (2000). Tyrosine kinase signalling in breast cancer: epidermal growth factor receptor and c-Src interactions in breast cancer. Breast Cancer Res 2: 203–210.
Biscardi JS, Maa MC, Tice DA, Cox ME, Leu TH, Parsons SJ . (1999a). c-Src-mediated phosphorylation of the epidermal growth factor receptor on Tyr845 and Tyr1101 is associated with modulation of receptor function. J Biol Chem 274: 8335–8343.
Biscardi JS, Tice DA, Parsons SJ . (1999b). c-Src, receptor tyrosine kinases, and human cancer. Adv Cancer Res 76: 61–119.
Bivona TG, Perez De Castro I, Ahearn IM, Grana TM, Chiu VK, Lockyer PJ et al. (2003). Phospholipase Cgamma activates Ras on the Golgi apparatus by means of RasGRP1. Nature 424: 694–698.
Blake RA, Garcia-Paramio P, Parker PJ, Courtneidge SA . (1999). Src promotes PKCdelta degradation. Cell Growth Differ 10: 231–241.
Blume-Jensen P, Hunter T . (2001). Oncogenic kinase signalling. Nature 411: 355–365.
Bolen JB, Veillette A, Schwartz AM, DeSeau V, Rosen N . (1987). Activation of pp60c-src protein kinase activity in human colon carcinoma. Proc Natl Acad Sci USA 84: 2251–2255.
Bowman T, Broome MA, Sinibaldi D, Wharton W, Pledger WJ, Sedivy JM et al. (2001). Stat3-mediated Myc expression is required for Src transformation and PDGF-induced mitogenesis. Proc Natl Acad Sci USA 98: 7319–7324.
Bromberg JF, Horvath CM, Besser D, Lathem WW, Darnell Jr JE . (1998). Stat3 activation is required for cellular transformation by v-src. Mol Cell Biol 18: 2553–2558.
Cappuzzo F, Finocchiaro G, Rossi E, Janne PA, Carnaghi C, Calandri C et al. (2008). EGFR FISH assay predicts for response to cetuximab in chemotherapy refractory colorectal cancer patients. Ann Oncol 19: 717–723.
Cartwright CA, Coad CA, Egbert BM . (1994). Elevated c-Src tyrosine kinase activity in premalignant epithelia of ulcerative colitis. J Clin Invest 93: 509–515.
Cartwright CA, Kamps MP, Meisler AI, Pipas JM, Eckhart W . (1989). pp60c-src activation in human colon carcinoma. J Clin Invest 83: 2025–2033.
Cartwright CA, Meisler AI, Eckhart W . (1990). Activation of the pp60c-src protein kinase is an early event in colonic carcinogenesis. Proc Natl Acad Sci USA 87: 558–562.
Chiu VK, Bivona T, Hach A, Sajous JB, Silletti J, Wiener H et al. (2002). Ras signalling on the endoplasmic reticulum and the Golgi. Nat Cell Biol 4: 343–350.
De Roock W, Piessevaux H, De Schutter J, Janssens M, De Hertogh G, Personeni N et al. (2008). KRAS wild-type state predicts survival and is associated to early radiological response in metastatic colorectal cancer treated with cetuximab. Ann Oncol 19: 508–515.
DeMali KA, Godwin SL, Soltoff SP, Kazlauskas A . (1999). Multiple roles for Src in a PDGF-stimulated cell. Exp Cell Res 253: 271–279.
Denning MF, Dlugosz AA, Threadgill DW, Magnuson T, Yuspa SH . (1996). Activation of the epidermal growth factor receptor signal transduction pathway stimulates tyrosine phosphorylation of protein kinase C delta. J Biol Chem 271: 5325–5331.
Di Fiore F, Blanchard F, Charbonnier F, Le Pessot F, Lamy A, Galais MP et al. (2007). Clinical relevance of KRAS mutation detection in metastatic colorectal cancer treated by Cetuximab plus chemotherapy. Br J Cancer 96: 1166–1169.
Emaduddin M, Bicknell DC, Bodmer WF, Feller SM . (2008). Cell growth, global phosphotyrosine elevation, and c-Met phosphorylation through Src family kinases in colorectal cancer cells. Proc Natl Acad Sci USA 105: 2358–2362.
Engelman JA . (2009). Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer 9: 550–562.
Fabian JR, Daar IO, Morrison DK . (1993). Critical tyrosine residues regulate the enzymatic and biological activity of Raf-1 kinase. Mol Cell Biol 13: 7170–7179.
Fearon ER, Vogelstein B . (1990). A genetic model for colorectal tumorigenesis. Cell 61: 759–767.
Fu YN, Yeh CL, Cheng HH, Yang CH, Tsai SF, Huang SF et al. (2008). EGFR mutants found in non-small cell lung cancer show different levels of sensitivity to suppression of Src: implications in targeting therapy. Oncogene 27: 957–965.
Ghoda L, Lin X, Greene WC . (1997). The 90-kDa ribosomal S6 kinase (pp90rsk) phosphorylates the N-terminal regulatory domain of IkappaBalpha and stimulates its degradation in vitro. J Biol Chem 272: 21281–21288.
Hantschel O, Rix U, Superti-Furga G . (2008). Target spectrum of the BCR-ABL inhibitors imatinib, nilotinib and dasatinib. Leuk Lymphoma 49: 615–619.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W et al. (2004). Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350: 2335–2342.
Irby R, Mao W, Coppola D, Jove R, Gamero A, Cuthbertson D et al. (1997). Overexpression of normal c-Src in poorly metastatic human colon cancer cells enhances primary tumor growth but not metastatic potential. Cell Growth Differ 8: 1287–1295.
Irby RB, Mao W, Coppola D, Kang J, Loubeau JM, Trudeau W et al. (1999). Activating SRC mutation in a subset of advanced human colon cancers. Nat Genet 21: 187–190.
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T et al. (2008). Cancer statistics, 2008. CA Cancer J Clin 58: 71–96.
Jhawer M, Goel S, Wilson AJ, Montagna C, Ling YH, Byun DS et al. (2008). PIK3CA mutation/PTEN expression status predicts response of colon cancer cells to the epidermal growth factor receptor inhibitor cetuximab. Cancer Res 68: 1953–1961.
Joseloff E, Cataisson C, Aamodt H, Ocheni H, Blumberg P, Kraker AJ et al. (2002). Src family kinases phosphorylate protein kinase C delta on tyrosine residues and modify the neoplastic phenotype of skin keratinocytes. J Biol Chem 277: 12318–12323.
Kantarjian H, Jabbour E, Grimley J, Kirkpatrick P . (2006). Dasatinib. Nat Rev Drug Discov 5: 717–718.
Karapetis CS, Khambata-Ford S, Jonker DJ, O'Callaghan CJ, Tu D, Tebbutt NC et al. (2008). K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N Engl J Med 359: 1757–1765.
Khambata-Ford S, Garrett CR, Meropol NJ, Basik M, Harbison CT, Wu S et al. (2007). Expression of epiregulin and amphiregulin and K-ras mutation status predict disease control in metastatic colorectal cancer patients treated with cetuximab. J Clin Oncol 25: 3230–3237.
Kijima T, Niwa H, Steinman RA, Drenning SD, Gooding WE, Wentzel AL et al. (2002). STAT3 activation abrogates growth factor dependence and contributes to head and neck squamous cell carcinoma tumor growth in vivo. Cell Growth Differ 13: 355–362.
Kim LC, Song L, Haura EB . (2009). Src kinases as therapeutic targets for cancer. Nat Rev Clin Oncol 6: 587–595.
Koppikar P, Choi SH, Egloff AM, Cai Q, Suzuki S, Freilino M et al. (2008). Combined inhibition of c-Src and epidermal growth factor receptor abrogates growth and invasion of head and neck squamous cell carcinoma. Clin Cancer Res 14: 4284–4291.
Kronfeld I, Kazimirsky G, Lorenzo PS, Garfield SH, Blumberg PM, Brodie C . (2000). Phosphorylation of protein kinase Cdelta on distinct tyrosine residues regulates specific cellular functions. J Biol Chem 275: 35491–35498.
Li C, Iida M, Dunn EF, Ghia AJ, Wheeler DL . (2009). Nuclear EGFR contributes to acquired resistance to cetuximab. Oncogene 28: 3801–3813.
Li W, Yu JC, Michieli P, Beeler JF, Ellmore N, Heidaran MA et al. (1994). Stimulation of the platelet-derived growth factor beta receptor signaling pathway activates protein kinase C-delta. Mol Cell Biol 14: 6727–6735.
Li Y, Kufe D . (2001). The human DF3/MUC1 carcinoma-associated antigen signals nuclear localization of the catenin p120(ctn). Biochem Biophys Res Commun 281: 440–443.
Li Y, Kuwahara H, Ren J, Wen G, Kufe D . (2001a). The c-Src tyrosine kinase regulates signaling of the human DF3/MUC1 carcinoma-associated antigen with GSK3 beta and beta-catenin. J Biol Chem 276: 6061–6064.
Li Y, Ren J, Yu W, Li Q, Kuwahara H, Yin L et al. (2001b). The epidermal growth factor receptor regulates interaction of the human DF3/MUC1 carcinoma antigen with c-Src and beta-catenin. J Biol Chem 276: 35239–35242.
Lievre A, Bachet JB, Le Corre D, Boige V, Landi B, Emile JF et al. (2006). KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res 66: 3992–3995.
Maa MC, Leu TH, McCarley DJ, Schatzman RC, Parsons SJ . (1995). Potentiation of epidermal growth factor receptor-mediated oncogenesis by c-Src: implications for the etiology of multiple human cancers. Proc Natl Acad Sci USA 92: 6981–6985.
Mao W, Irby R, Coppola D, Fu L, Wloch M, Turner J et al. (1997). Activation of c-Src by receptor tyrosine kinases in human colon cancer cells with high metastatic potential. Oncogene 15: 3083–3090.
Marmor MD, Skaria KB, Yarden Y . (2004). Signal transduction and oncogenesis by ErbB/HER receptors. Int J Radiat Oncol Biol Phys 58: 903–913.
McLean GW, Carragher NO, Avizienyte E, Evans J, Brunton VG, Frame MC . (2005). The role of focal-adhesion kinase in cancer—a new therapeutic opportunity. Nat Rev Cancer 5: 505–515.
Muthuswamy SK, Muller WJ . (1995). Direct and specific interaction of c-Src with Neu is involved in signaling by the epidermal growth factor receptor. Oncogene 11: 271–279.
Muthuswamy SK, Siegel PM, Dankort DL, Webster MA, Muller WJ . (1994). Mammary tumors expressing the neu proto-oncogene possess elevated c-Src tyrosine kinase activity. Mol Cell Biol 14: 735–743.
Normanno N, Bianco C, De Luca A, Maiello MR, Salomon DS . (2003). Target-based agents against ErbB receptors and their ligands: a novel approach to cancer treatment. Endocr Relat Cancer 10: 1–21.
Normanno N, Tejpar S, Morgillo F, De Luca A, Van Cutsem E, Ciardiello F . (2009). Implications for KRAS status and EGFR-targeted therapies in metastatic CRC. Nat Rev Clin Oncol 6: 519–527.
Prenzel N, Fischer OM, Streit S, Hart S, Ullrich A . (2001). The epidermal growth factor receptor family as a central element for cellular signal transduction and diversification. Endocr Relat Cancer 8: 11–31.
Richards SA, Fu J, Romanelli A, Shimamura A, Blenis J . (1999). Ribosomal S6 kinase 1 (RSK1) activation requires signals dependent on and independent of the MAP kinase ERK. Curr Biol 9: 810–820.
Rini BI . (2008). Temsirolimus, an inhibitor of mammalian target of rapamycin. Clin Cancer Res 14: 1286–1290.
Schaller MD, Hildebrand JD, Shannon JD, Fox JW, Vines RR, Parsons JT . (1994). Autophosphorylation of the focal adhesion kinase, pp125FAK, directs SH2-dependent binding of pp60src. Mol Cell Biol 14: 1680–1688.
Schlessinger J . (2000). Cell signaling by receptor tyrosine kinases. Cell 103: 211–225.
Shimamura A, Ballif BA, Richards SA, Blenis J . (2000). Rsk1 mediates a MEK-MAP kinase cell survival signal. Curr Biol 10: 127–135.
Summy JM, Gallick GE . (2003). Src family kinases in tumor progression and metastasis. Cancer Metastasis Rev 22: 337–358.
Talamonti MS, Roh MS, Curley SA, Gallick GE . (1993). Increase in activity and level of pp60c-src in progressive stages of human colorectal cancer. J Clin Invest 91: 53–60.
Termuhlen PM, Curley SA, Talamonti MS, Saboorian MH, Gallick GE . (1993). Site-specific differences in pp60c-src activity in human colorectal metastases. J Surg Res 54: 293–298.
Tice DA, Biscardi JS, Nickles AL, Parsons SJ . (1999). Mechanism of biological synergy between cellular Src and epidermal growth factor receptor. Proc Natl Acad Sci USA 96: 1415–1420.
Viloria-Petit A, Crombet T, Jothy S, Hicklin D, Bohlen P, Schlaeppi JM et al. (2001). Acquired resistance to the antitumor effect of epidermal growth factor receptor-blocking antibodies in vivo: a role for altered tumor angiogenesis. Cancer Res 61: 5090–5101.
Wee S, Jagani Z, Xiang KX, Loo A, Dorsch M, Yao YM et al. (2009). PI3K pathway activation mediates resistance to MEK inhibitors in KRAS mutant cancers. Cancer Res 69: 4286–4293.
Wheeler DL, Huang S, Kruser TJ, Nechrebecki MM, Armstrong EA, Benavente S et al. (2008). Mechanisms of acquired resistance to cetuximab: role of HER (ErbB) family members. Oncogene 27: 3944–3956.
Wheeler DL, Iida M, Kruser TJ, Nechrebecki MM, Dunn EF, Armstrong EA et al. (2009). Epidermal growth factor receptor cooperates with Src family kinases in acquired resistance to cetuximab. Cancer Biol Ther 8: 696–703.
Wiggin GR, Soloaga A, Foster JM, Murray-Tait V, Cohen P, Arthur JS . (2002). MSK1 and MSK2 are required for the mitogen- and stress-induced phosphorylation of CREB and ATF1 in fibroblasts. Mol Cell Biol 22: 2871–2881.
Xu W, Doshi A, Lei M, Eck MJ, Harrison SC . (1999). Crystal structures of c-Src reveal features of its autoinhibitory mechanism. Mol Cell 3: 629–638.
Yarden Y, Sliwkowski MX . (2001). Untangling the ErbB signalling network. Nat Rev Mol Cell Biol 2: 127–137.
Yu CL, Meyer DJ, Campbell GS, Larner AC, Carter-Su C, Schwartz J et al. (1995). Enhanced DNA-binding activity of a Stat3-related protein in cells transformed by the Src oncoprotein. Science 269: 81–83.
Zhang J, Kalyankrishna S, Wislez M, Thilaganathan N, Saigal B, Wei W et al. (2007). SRC-family kinases are activated in non-small cell lung cancer and promote the survival of epidermal growth factor receptor-dependent cell lines. Am J Pathol 170: 366–376.
Acknowledgements
This project was supported, in part, by grant P30CA014520 from the National Cancer Institute, grant 1UL1RR025011 from the Clinical and Translational Science Award program of the National Center for Research Resources, National Institutes of Health (to DLW), by grant RSG-10-193-01-TBG from the American Cancer Society (to DLW) and T32 grant CA009614-17 Physician Scientist Training in Cancer Medicine from the National Institutes of Health (to EFD).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
DLW has held a sponsored research agreement with Bristol-Myers Squibb.
Rights and permissions
About this article
Cite this article
Dunn, E., Iida, M., Myers, R. et al. Dasatinib sensitizes KRAS mutant colorectal tumors to cetuximab. Oncogene 30, 561–574 (2011). https://doi.org/10.1038/onc.2010.430
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.430
Keywords
This article is cited by
-
Platycodin D represses β-catenin to suppress metastasis of cetuximab-treated KRAS wild-type colorectal cancer cells
Clinical & Experimental Metastasis (2023)
-
Promotion or remission: a role of noncoding RNAs in colorectal cancer resistance to anti-EGFR therapy
Cell Communication and Signaling (2022)
-
Activation of FXR and inhibition of EZH2 synergistically inhibit colorectal cancer through cooperatively accelerating FXR nuclear location and upregulating CDX2 expression
Cell Death & Disease (2022)
-
Src family kinases, adaptor proteins and the actin cytoskeleton in epithelial-to-mesenchymal transition
Cell Communication and Signaling (2021)
-
Resistance to anti-EGFR therapies in metastatic colorectal cancer: underlying mechanisms and reversal strategies
Journal of Experimental & Clinical Cancer Research (2021)