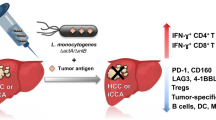
Abstract
Live attenuated Listeria monocytogenes (LM) is a promising bacterial vector able to induce a T-cell response to tumor-associated antigens and demonstrates great potential for use in vaccine development. A novel recombinant LM-based vaccine (Lmdd (LM ΔdalΔdat)-MPFG (multiple peptide fusing genes)) was developed with the ability to express and secrete hepatocellular carcinoma (HCC)-related tumor-associated antigens fragments due to the insertion of hepatitis B virus (HBV)-X protein (HBx)-derived epitopes HBx52−60 and HBx140−148, the universal T-helper epitope, alpha-fetoprotein (AFP) epitope AFP158−166, and melanoma antigen gene (MAGE)-3271−279 into the HBV core protein. Following immunization with the Lmdd-MPFG vaccine, macrophages exhibited uptake of the bacteria; the vaccine was then nearly cleared 3 days after the first administration. It disappeared even more quickly following subsequent vaccinations. However, recombinant Lmdd-MPFG allowed for the full development of an antitumor response towards the human leukocyte antigen (HLA)-A0201 epitopes of MPFG. Each epitope stimulated an augmented T-cell proliferation and enhanced the supernatant level of interferon (IFN)-γ in vitro. In addition, IFN-γ-producing CD8+ T cells as well as in vivo cytolytic activity were significantly increased in HLA-A2 transgenic mice. Additionally, the Lmdd-MPFG developed a strong antitumor response, as indicated by the significant resistance of immunized mice to MPFG-positive Hepa1-6 cell challenge in both a prophylactic and therapeutic setting. Tumor regression was accompanied by an enhanced cytotoxic T lymphocyte response and a decrease of regulatory T cells in the tumor. Collectively, these results suggest that utilizing attenuated LM as a vaccine vector, able to carry the MPFG gene, presents a potentially feasible strategy for prevention of HCC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Alouf JE . (2000). Cholesterol-binding cytolytic protein toxins. Int J Med Microbiol 290: 351–356.
Badvie S . (2000). Hepatocellular carcinoma. Postgrad Med J 76: 4–11.
Bai X, Zhu Y, Jin Y, Guo X, Qian G, Chen T et al. (2011). Temporal acquisition of sequential mutations in the enhancer II and basal core promoter of HBV in individuals at high risk for hepatocellular carcinoma. Carcinogenesis 32: 63–68.
Bertoletti A, Chisari FV, Penna A, Guilhot S, Galati L, Missale G et al. (1993). Definition of a minimal optimal cytotoxic T-cell epitope within the hepatitis B virus nucleocapsid protein. J Virol 67: 2376–2380.
Blasco H, Lalmanach G, Godat E, Maurel MC, Canepa S, Belghazi M et al. (2007). Evaluation of a peptide ELISA for the detection of rituximab in serum. J Immunol Methods 325: 127–139.
Butterfield LH, Koh A, Meng W, Vollmer CM, Ribas A, Dissette V et al. (1999). Generation of human T-cell responses to an HLA-A2.1-restricted peptide epitope derived from alpha-fetoprotein. Cancer Res 59: 3134–3142.
Butterfield LH, Meng WS, Koh A, Vollmer CM, Ribas A, Dissette VB et al. (2001). T cell responses to HLA-A*0201-restricted peptides derived from human alpha fetoprotein. J Immunol 166: 5300–5308.
Chen Y, Sun H, Liu G, Wang B, Wang F, Sun B et al. (2009). EBV LMP2A-specific T cell immune responses elicited by dendritic cells loaded with LMP2A protein. Cell Mol Immunol 6: 269–276.
Cheng Z, Hu J, King J, Jay G, Campbell TC . (1997). Inhibition of hepatocellular carcinoma development in hepatitis B virus transfected mice by low dietary casein. Hepatology 26: 1351–1354.
Ding FX, Wang F, Lu YM, Li K, Wang KH, He XW et al. (2009). Multiepitope peptide-loaded virus-like particles as a vaccine against hepatitis B virus-related hepatocellular carcinoma. Hepatology 49: 1492–1502.
Duvoux C . (1998). [Epidemiology and diagnosis of hepatocellular carcinomas in cirrhosis]. Ann Chir 52: 511–517.
Frankel FR, Hegde S, Lieberman J, Paterson Y . (1995). Induction of cell-mediated immune responses to human immunodeficiency virus type 1 Gag protein by using Listeria monocytogenes as a live vaccine vector. J Immunol 155: 4775–4782.
Friedman RS, Frankel FR, Xu Z, Lieberman J . (2000). Induction of human immunodeficiency virus (HIV)-specific CD8 T-cell responses by Listeria monocytogenes and a hyperattenuated Listeria strain engineered to express HIV antigens. J Virol 74: 9987–9993.
Furukawa S, Akbar SM, Hasebe A, Horiike N, Onji M . (2004). Production of hepatitis B surface antigen-pulsed dendritic cells from immunosuppressed murine hepatitis B virus carrier: evaluation of immunogenicity of antigen-pulsed dendritic cells in vivo. Immunobiology 209: 551–557.
Gedde MM, Higgins DE, Tilney LG, Portnoy DA . (2000). Role of listeriolysin O in cell-to-cell spread of Listeria monocytogenes. Infect Immun 68: 999–1003.
Greten TF, Ormandy LA, Fikuart A, Hochst B, Henschen S, Horning M et al. (2010). Low-dose cyclophosphamide treatment impairs regulatory T cells and unmasks AFP-specific CD4+ T-cell responses in patients with advanced HCC. J Immunother 33: 211–218.
Hussain SA, Ferry DR, El-Gazzaz G, Mirza DF, James ND, McMaster P et al. (2001). Hepatocellular carcinoma. Ann Oncol 12: 161–172.
Kim CM, Koike K, Saito I, Miyamura T, Jay G . (1991). HBx gene of hepatitis B virus induces liver cancer in transgenic mice. Nature 351: 317–320.
Kong J, Diao Z, Deng X, Zhong H, Yao W, Hu X . (2007). Anti-tumor effects of immunotherapeutic peptide on the treatment of hepatocellular carcinoma with HBc carrier. Oncol Rep 18: 279–285.
Kursar M, Bonhagen K, Kohler A, Kamradt T, Kaufmann SH, Mittrucker HW . (2002). Organ-specific CD4+ T cell response during Listeria monocytogenes infection. J Immunol 168: 6382–6387.
Li Z, Zhang M, Zhou C, Zhao X, Iijima N, Frankel FR . (2008). Novel vaccination protocol with two live mucosal vectors elicits strong cell-mediated immunity in the vagina and protects against vaginal virus challenge. J Immunol 180: 2504–2513.
Lin SM, Lin CJ, Lin CC, Hsu CW, Chen YC . (2005). Randomised controlled trial comparing percutaneous radiofrequency thermal ablation, percutaneous ethanol injection, and percutaneous acetic acid injection to treat hepatocellular carcinoma of 3 cm or less. Gut 54: 1151–1156.
Mackaness GB . (1969). The influence of immunologically committed lymphoid cells on macrophage activity in vivo. J Exp Med 129: 973–992.
Menetrier-Caux C, Gobert M, Caux C . (2009). Differences in tumor regulatory T-cell localization and activation status impact patient outcome. Cancer Res 69: 7895–7898.
Nieters A, Yuan JM, Sun CL, Zhang ZQ, Stoehlmacher J, Govindarajan S et al. (2005). Effect of cytokine genotypes on the hepatitis B virus-hepatocellular carcinoma association. Cancer 103: 740–748.
Olkhanud PB, Baatar D, Bodogai M, Hakim F, Gress R, Anderson RL et al. (2009). Breast cancer lung metastasis requires expression of chemokine receptor CCR4 and regulatory T cells. Cancer Res 69: 5996–6004.
Pamer EG . (2004). Immune responses to Listeria monocytogenes. Nat Rev Immunol 4: 812–823.
Pamer EG, Sijts AJ, Villanueva MS, Busch DH, Vijh S . (1997). MHC class I antigen processing of Listeria monocytogenes proteins: implications for dominant and subdominant CTL responses. Immunol Rev 158: 129–136.
Peters C, Paterson Y . (2003). Enhancing the immunogenicity of bioengineered Listeria monocytogenes by passaging through live animal hosts. Vaccine 21: 1187–1194.
Ritter M, Ali MY, Grimm CF, Weth R, Mohr L, Bocher WO et al. (2004). Immunoregulation of dendritic and T cells by alpha-fetoprotein in patients with hepatocellular carcinoma. J Hepatol 41: 999–1007.
Sanyal AJ, Yoon SK, Lencioni R . (2010). The etiology of hepatocellular carcinoma and consequences for treatment. Oncologist 15 (Suppl 4): 14–22.
Sciaranghella G, Lakhashe SK, Ayash-Rashkovsky M, Mirshahidi S, Siddappa NB, Novembre FJ et al. (2011). A live attenuated Listeria monocytogenes vaccine vector expressing SIV Gag is safe and immunogenic in macaques and can be administered repeatedly. Vaccine 29: 476–486.
Seavey MM, Pan ZK, Maciag PC, Wallecha A, Rivera S, Paterson Y et al. (2009). A novel human Her-2/neu chimeric molecule expressed by Listeria monocytogenes can elicit potent HLA-A2 restricted CD8-positive T cell responses and impact the growth and spread of Her-2/neu-positive breast tumors. Clin Cancer Res 15: 924–932.
Shedlock DJ, Whitmire JK, Tan J, MacDonald AS, Ahmed R, Shen H . (2003). Role of CD4T cell help and costimulation in CD8T cell responses during Listeria monocytogenes infection. J Immunol 170: 2053–2063.
Shen F, Chen PD, Walfield AM, Ye J, House J, Brown F et al. (1999). Differentiation of convalescent animals from those vaccinated against foot-and-mouth disease by a peptide ELISA. Vaccine 17: 3039–3049.
Shen X, Li N, Li H, Zhang T, Wang F, Li Q et al. (2010a). Increased prevalence of regulatory T cells in the tumor microenvironment and its correlation with TNM stage of hepatocellular carcinoma. J Cancer Res Clin Oncol 136: 1745–1754.
Shen Y, Kawamura I, Nomura T, Tsuchiya K, Hara H, Dewamitta SR et al. (2010b). Toll-like receptor 2- and MyD88-dependent phosphatidylinositol 3-kinase and Rac1 activation facilitates the phagocytosis of Listeria monocytogenes by murine macrophages. Infect Immun 78: 2857–2867.
Singh R, Paterson Y . (2006). Listeria monocytogenes as a vector for tumor-associated antigens for cancer immunotherapy. Expert Rev Vaccines 5: 541–552.
Skoberne M, Yewdall A, Bahjat KS, Godefroy E, Lauer P, Lemmens E et al. (2008). KBMA Listeria monocytogenes is an effective vector for DC-mediated induction of antitumor immunity. J Clin Invest 118: 3990–4001.
Smith K, Youngman P . (1992). Use of a new integrational vector to investigate compartment-specific expression of the Bacillus subtilis spoIIM gene. Biochimie 74: 705–711.
Song S, Wang F, He X, He Y, Li D, Sun S . (2007). Evaluation of antitumor immunity efficacy of epitope-based vaccine with B16 cell line coexpressing HLA-A2/H-2 kb and CTL multiepitope in HLA transgenic mice. Vaccine 25: 4853–4860.
Souders NC, Verch T, Paterson Y . (2006). in vivo bactofection: listeria can function as a DNA-cancer vaccine. DNA Cell Biol 25: 142–151.
Spears PA, Suyemoto MM, Palermo AM, Horton JR, Hamrick TS, Havell EA et al. (2008). A Listeria monocytogenes mutant defective in bacteriophage attachment is attenuated in orally inoculated mice and impaired in enterocyte intracellular growth. Infect Immun 76: 4046–4054.
Su F, Schneider RJ . (1997). Hepatitis B virus HBx protein sensitizes cells to apoptotic killing by tumor necrosis factor alpha. Proc Natl Acad Sci USA 94: 8744–8749.
Su Q, Schroder CH, Hofmann WJ, Otto G, Pichlmayr R, Bannasch P . (1998). Expression of hepatitis B virus X protein in HBV-infected human livers and hepatocellular carcinomas. Hepatology 27: 1109–1120.
Thompson RJ, Bouwer HG, Portnoy DA, Frankel FR . (1998). Pathogenicity and immunogenicity of a Listeria monocytogenes strain that requires D-alanine for growth. Infect Immun 66: 3552–3561.
Vollmer Jr CM, Eilber FC, Butterfield LH, Ribas A, Dissette VB, Koh A et al. (1999). Alpha-fetoprotein-specific genetic immunotherapy for hepatocellular carcinoma. Cancer Res 59: 3064–3067.
Wallecha A, Maciag PC, Rivera S, Paterson Y, Shahabi V . (2009). Construction and characterization of an attenuated Listeria monocytogenes strain for clinical use in cancer immunotherapy. Clin Vaccine Immunol 16: 96–103.
Williams JC, Wagner NJ, Earp HS, Vilen BJ, Matsushima GK . (2010). Increased hematopoietic cells in the mertk-/- mouse peritoneal cavity: a result of augmented migration. J Immunol 184: 6637–6648.
Yamamoto K, Kawamura I, Tominaga T, Nomura T, Ito J, Mitsuyama M . (2006). Listeriolysin O derived from Listeria monocytogenes inhibits the effector phase of an experimental allergic rhinitis induced by ovalbumin in mice. Clin Exp Immunol 144: 475–484.
Yoshimura K, Jain A, Allen HE, Laird LS, Chia CY, Ravi S et al. (2006). Selective targeting of antitumor immune responses with engineered live-attenuated Listeria monocytogenes. Cancer Res 66: 1096–1104.
Yoshimura K, Laird LS, Chia CY, Meckel KF, Slansky JE, Thompson JM et al. (2007). Live attenuated Listeria monocytogenes effectively treats hepatic colorectal cancer metastases and is strongly enhanced by depletion of regulatory T cells. Cancer Res 67: 10058–10066.
Zerbini A, Pilli M, Soliani P, Ziegler S, Pelosi G, Orlandini A et al. (2004). Ex vivo characterization of tumor-derived melanoma antigen encoding gene-specific CD8+cells in patients with hepatocellular carcinoma. J Hepatol 40: 102–109.
Zhao X, Li Z, Gu B, Frankel FR . (2005). Pathogenicity and immunogenicity of a vaccine strain of Listeria monocytogenes that relies on a suicide plasmid to supply an essential gene product. Infect Immun 73: 5789–5798.
Zhou L, Fu JL, Lu YY, Fu BY, Wang CP, An LJ et al. (2010). Regulatory T cells are associated with post-cryoablation prognosis in patients with hepatitis B virus-related hepatocellular carcinoma. J Gastroenterol 45: 968–978.
Zhou M, Peng JR, Zhang HG, Wang HX, Zhong ZH, Pan XY et al. (2005). Identification of two naturally presented MAGE antigenic peptides from a patient with hepatocellular carcinoma by mass spectrometry. Immunol Lett 99: 113–121.
Acknowledgements
This work was supported by grants from National Basic Research Program of China (2012CB910800 to BS), the National Natural Science Foundation (81072029 to BS and 30901750 to YC), Major Research Plan of the National Natural Science Foundation (91029721 to BS), the New Century Excellent Talents in University, Ministry of Education (NCET-09-0160 to BS), Natural Science Foundation of Jiangsu Province (BK2010532 to YC), Ministry of Health, China (W201001 to LD), and National Institutes of Health grant (R21-AI-071774 to FRF).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Chen, Y., Yang, D., Li, S. et al. Development of a Listeria monocytogenes-based vaccine against hepatocellular carcinoma. Oncogene 31, 2140–2152 (2012). https://doi.org/10.1038/onc.2011.395
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.395
Keywords
This article is cited by
-
Roles of tumor-associated macrophages in anti-PD-1/PD-L1 immunotherapy for solid cancers
Molecular Cancer (2023)
-
Safety and efficacy of prophylactic and therapeutic vaccine based on live-attenuated Listeria monocytogenes in hepatobiliary cancers
Oncogene (2022)
-
Identification of HLA-A2 restricted epitopes of glypican-3 and induction of CTL responses in HLA-A2 transgenic mice
Cancer Immunology, Immunotherapy (2022)
-
Targeting tumor-associated macrophages to synergize tumor immunotherapy
Signal Transduction and Targeted Therapy (2021)
-
Protective CD8+ T-cell response against Hantaan virus infection induced by immunization with designed linear multi-epitope peptides in HLA-A2.1/Kb transgenic mice
Virology Journal (2020)