Abstract
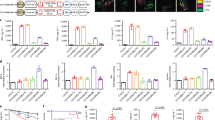
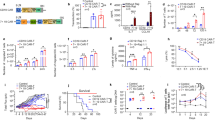
Anti-CD19 chimeric antigen receptor (CAR)-expressing T cells are an effective treatment for B-cell lymphoma, but often cause neurologic toxicity. We treated 20 patients with B-cell lymphoma on a phase I, first-in-human clinical trial of T cells expressing the new anti-CD19 CAR Hu19-CD828Z (NCT02659943). The primary objective was to assess safety and feasibility of Hu19-CD828Z T-cell therapy. Secondary objectives included assessments of blood levels of CAR T cells, anti-lymphoma activity, second infusions and immunogenicity. All objectives were met. Fifty-five percent of patients who received Hu19-CD828Z T cells obtained complete remission. Hu19-CD828Z T cells had clinical anti-lymphoma activity similar to that of T cells expressing FMC63-28Z, an anti-CD19 CAR tested previously by our group, which contains murine binding domains and is used in axicabtagene ciloleucel. However, severe neurologic toxicity occurred in only 5% of patients who received Hu19-CD828Z T cells, whereas 50% of patients who received FMC63-28Z T cells experienced this degree of toxicity (P = 0.0017). T cells expressing Hu19-CD828Z released lower levels of cytokines than T cells expressing FMC63-28Z. Lower levels of cytokines were detected in blood from patients who received Hu19-CD828Z T cells than in blood from those who received FMC63-28Z T cells, which could explain the lower level of neurologic toxicity associated with Hu19-CD828Z. Levels of cytokines released by CAR-expressing T cells particularly depended on the hinge and transmembrane domains included in the CAR design.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All requests for raw and analyzed data and materials are promptly reviewed by the National Cancer Institute Technology Transfer Center to verify whether the request is subject to any intellectual property or confidentiality obligations. Patient-related data not included in the paper were generated as part of clinical trials and may be subject to patient confidentiality. Any data and materials that can be shared will be released via a material transfer agreement. All other data that support the findings of this study will be provided by the corresponding author upon reasonable request when possible. Raw data for all Figs. 1–5 and Extended Data Fig. 3 are in the submitted Source Data Excel file.
CAR sequences were all submitted to GenBank.
GenBank accession number for LSIN-Hu19-CD828Z: MN698642
GenBank accession number for LSIN-FMC63-CD828Z: MN702884
GenBank accession number for LSIN-Hu19-28Z: MN702882
GenBank accession number for MSGV-Hu19-CD828Z: MN702883
GenBank accession number for MSGV-FMC63-28Z: HM852952.1
Change history
14 April 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41591-020-0864-x
References
Turtle, C. J. et al. Immunotherapy of non-Hodgkin’s lymphoma with a defined ratio of CD8+ and CD4+ CD19-specific chimeric antigen receptor-modified T cells. Sci. Transl. Med 8, 355ra116 (2016).
Kochenderfer, J. N. et al. Eradication of B-lineage cells and regression of lymphoma in a subject treated with autologous T cells genetically engineered to recognize CD19. Blood 116, 4099–4102 (2010).
Kochenderfer, J. N. et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood 119, 2709–2720 (2012).
Kochenderfer, J. N. et al. Chemotherapy-refractory diffuse large B-cell lymphoma and indolent B-cell malignancies can be effectively treated with autologous T cells expressing an anti-CD19 chimeric antigen receptor. J. Clin. Oncol. 33, 540–549 (2015).
Kochenderfer, J. N. et al. Lymphoma remissions caused by anti-CD19 chimeric antigen receptor T cells are associated with high serum interleukin-15 levels. J. Clin. Oncol. 35, 1803–1813 (2017).
Kochenderfer, J. N. et al. Long-duration complete remissions of diffuse large B cell lymphoma after anti-CD19 chimeric antigen receptor T cell therapy. Mol. Ther. 25, 2245–2253 (2017).
Neelapu, S. S. et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Schuster, S. J. et al. Chimeric antigen receptor T cells in refractory B-cell lymphomas. N. Engl. J. Med. 377, 2545–2554 (2017).
Brudno, J. N. & Kochenderfer, J. N. Chimeric antigen receptor T-cell therapies for lymphoma. Nat. Rev. Clin. Oncol. 15, 31–46 (2018).
Savoldo, B. et al. CD28 costimulation improves expansion and persistence of chimeric antigen receptor-modified T cells in lymphoma subjects. J. Clin. Invest. 121, 1822–1826 (2011).
Schuster, S. J. et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N. Engl. J. Med. 380, 45–56 (2019).
Sadelain, M. CAR therapy: the CD19 paradigm. J. Clin. Invest. 125, 3392–3400 (2015).
Johnson, L. A. & June, C. H. Driving gene-engineered T cell immunotherapy of cancer. Cell Res. 27, 38–58 (2017).
Ramos, C. A., Heslop, H. E. & Brenner, M. K. CAR-T cell therapy for lymphoma. Annu. Rev. Med. 67, 165–183 (2016).
Ramos, C. A. et al. In vivo fate and activity of second- versus third-generation CD19-specific CAR-T cells in B cell non-Hodgkin’s lymphomas. Mol. Ther. 26, 2727–2737 (2018).
Locke, F. L. et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1–2 trial. Lancet. Oncol. 20, 31–42 (2019).
Brudno, J. N. & Kochenderfer, J. N. Recent advances in CAR T-cell toxicity: mechanisms, manifestations and management. Blood. Rev. 34, 45–55 (2019).
Brudno, J. N. & Kochenderfer, J. N. Toxicities of chimeric antigen receptor T cells: recognition and management. Blood 127, 3321–3330 (2016).
Neelapu, S. S. et al. Chimeric antigen receptor T-cell therapy-assessment and management of toxicities. Nat. Rev. Clin. Oncol. 15, 47–62 (2018).
Hirayama, A. V. & Turtle, C. J. Toxicities of CD19 CAR-T cell immunotherapy. Am. J. Hematol. 94, S42–S49 (2019).
Teachey, D. T. et al. Identification of predictive biomarkers for cytokine release syndrome after chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Cancer Discov 6, 664–679 (2016).
Singh, N. et al. Monocyte lineage–derived IL-6 does not affect chimeric antigen receptor T-cell function. Cytotherapy. 19, 867–880 (2017).
Santomasso, B. D. et al. Clinical and biological correlates of neurotoxicity associated with car t-cell therapy in subjects with B-cell acute lymphoblastic leukemia. Cancer Discov 8, 958–971 (2018).
Gust, J. et al. Endothelial activation and blood–brain barrier disruption in neurotoxicity after adoptive immunotherapy with CD19 CAR-T cells. Cancer Discov 7, 1404–1419 (2017).
Karschnia, P. et al. Clinical presentation, management, and biomarkers of neurotoxicity after adoptive immunotherapy with CAR T cells. Blood 133, 2212–2221 (2019).
Gust, J. et al. Glial injury in neurotoxicity after pediatric CD19-directed chimeric antigen receptor T cell therapy. Ann. Neurol. 86, 42–54 (2019).
Taraseviciute, A. et al. Chimeric antigen receptor T cell–mediated neurotoxicity in nonhuman primates. Cancer Discov 8, 750–763 (2018).
Alabanza, L. et al. Function of novel Anti-CD19 chimeric antigen receptors with human variable regions is affected by hinge and transmembrane domains. Mol. Ther. 25, 2452–2465 (2017).
Kochenderfer, J. N. et al. Construction and preclinical evaluation of an anti-CD19 chimeric antigen receptor. J. Immunother. 32, 689–702 (2009).
Hughes, M. S. et al. Transfer of a TCR gene derived from a subject with a marked antitumor response conveys highly active T-cell effector functions. Hum. Gene Ther. 16, 457–472 (2005).
Goff, S. L. et al. Randomized, prospective evaluation comparing intensity of lymphodepletion before adoptive transfer of tumor-infiltrating lymphocytes for subjects with metastatic melanoma. J. Clin. Oncol. 34, 2389–2397 (2016).
Kochenderfer, J. N., Yu, Z., Frasheri, D., Restifo, N. P. & Rosenberg, S. A. Adoptive transfer of syngeneic T cells transduced with a chimeric antigen receptor that recognizes murine CD19 can eradicate lymphoma and normal B cells. Blood 116, 3875–3886 (2010).
Gattinoni, L. et al. Removal of homeostatic cytokine sinks by lymphodepletion enhances the efficacy of adoptively transferred tumor-specific CD8+ T cells. J. Exp. Med. 202, 907–912 (2005).
North, R. J. Cyclophosphamide-facilitated adoptive immunotherapy of an established tumor depends on elimination of tumor-induced suppressor T cells. J. Exp. Med. 155, 1063–1074 (1982).
Sievers, S. A., Kelley, K. A., Astrow, S. H., Bot, A. & Wiltzius, J. J. W. Design and development of anti-linker antibodies for the detection and characterization of CAR T cells. Proceedings: AACR Annual Meeting, abstr. 1204 (2019).
Brudno, J. N. et al. Allogeneic T cells that express an anti-CD19 chimeric antigen receptor induce remissions of B-cell malignancies that progress after allogeneic hematopoietic stem-cell transplantation without causing graft-versus-host disease. J. Clin. Oncol. 34, 1112–1121 (2016).
Brudno, J. N. et al. T cells genetically modified to express an anti-B-Cell maturation antigen chimeric antigen receptor cause remissions of poor-prognosis relapsed multiple myeloma. J. Clin. Oncol. 36, 2267–2280 (2018).
Cheson, B. D. et al. Revised response criteria for malignant lymphoma. J. Clin. Oncol. 25, 579–586 (2007).
Lee, D. W. et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol. Blood. Marrow Transplant. 25, 625–638 (2019).
National Cancer Institute Common Terminology Criteria for Adverse Events v3.0 https://www.eortc.be/services/doc/ctc/ctcaev3.pdf (2006).
Sallusto, F., Lenig, D., Förster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Meierhoff, G., Ott, P. A., Lehmann, P. V. & Schloot, N. C. Cytokine detection by ELISPOT: relevance for immunological studies in type 1 diabetes. Diabetes Metab. Res. Rev. 18, 367–380 (2002).
De Angelo, D. et al. Clinical Outcomes for a phase II, single arm, multicenter trial of JCAR015 in adult B-ALL (ROCKET Study). SITC 2017 Annual Meeting Abstract Book, abstr. P217 (2017).
Brentjens, R. J. et al. Genetically targeted T cells eradicate systemic acute lymphoblastic leukemia xenografts. Clin. Cancer Res. 13, 5426–5435 (2007).
Ying, Z. et al. A safe and potent anti-CD19 CAR T cell therapy. Nat. Med. 25, 947–953 (2019).
Feucht, J. et al. Calibration of CAR activation potential directs alternative T cell fates and therapeutic potency. Nat. Med. 25, 82–88 (2019).
Ghorashian, S. et al. Enhanced CAR T cell expansion and prolonged persistence in pediatric subjects with ALL treated with a low-affinity CD19 CAR. Nat. Med. 25, 1408–1414 (2019).
Zheng, Z., Chinnasamy, N. & Morgan, R. A. Protein L: a novel reagent for the detection of chimeric antigen receptor (CAR) expression by flow cytometry. J. Transl. Med. 10, 29 (2012).
Feldman, S. A. et al. Rapid production of clinical-grade gammaretroviral vectors in expanded surface roller bottles using a “modified” step-filtration process for clearance of packaging cells. Hum. Gene Ther. 22, 107–115 (2011).
Warren, E. H. et al. Therapy of relapsed leukemia after allogeneic hematopoietic cell transplantation with T cells specific for minor histocompatibility antigens. Blood 115, 3869–3878 (2010).
Morgan, R. A. et al. Case report of a serious adverse event following the administration of t cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol. Ther. 18, 843–851 (2010).
Cachau, R. E., Erickson, J. W. & Villar, H. O. Novel procedure for structure refinement in homology modeling and its application to the human class mu glutathione s-transferases. Protein Eng. Des. Sel. 7, 831–839 (1994).
Yokoyama, S. et al. A novel pathway of LPS uptake through syndecan-1 leading to pyroptotic cell death. eLife 7, e37854 (2018).
Marrink, S. J. et al. Computational modeling of realistic cell membranes. Chem. Rev. 119, 6184–6226 (2019).
Bello, M. & Correa-Basurto, J. Energetic and flexibility properties captured by long molecular dynamics simulations of a membrane-embedded pMHCII-TCR complex. Mol. Biosyst. 12, 1350–1366 (2016).
Acknowledgements
The clinical LSIN-Hu19-CD828Z vector was produced and gene-therapy monitoring was performed with assistance from the NHLBI-funded National Gene Vector Biorepository at Indiana University. We thank the following clinical units at the NIH clinical center for patient care: Experimental Transplantation and Immunotherapy Branch Clinical Service, the 3 Northeast Nursing Unit and the Department of Critical Care Medicine and the Intensive Care Unit staff. Funding for this work was from National Cancer Institute Intramural funding and Kite, a Gilead Company.
Author information
Authors and Affiliations
Contributions
J.N.K. designed the trial and supervised all activities. J.N.B. and J.N.K. wrote the paper. J.N.B., N.L., D.V., J.J.R., Y.-w.S., J.R., A.X., A.B., N.S., L.M., R.C., P.Y., B.H., D.F.S., S.A.R. and J.N.K. analyzed data. J.N.B., L.M., R.E., M.R., R.D., B.H., R.P., R.E.G. and J.N.K. provided patient care. N.L., D.V., Y-W.S., J.J.R., R.C. and P.Y. conducted experiments. All authors reviewed, edited and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
This work was supported by intramural funding of the Center for Cancer Research, NCI, NIH. In addition, the NCI has cooperative research and development agreements with Kite Pharma, a Gilead Company that supports development of anti-CD19 CAR T-cell therapies, and both J.N.K. and S.A.R. are NCI principal investigators of these research agreements. J.K. has a patent application for the Hu19-CD828Z CAR and has received royalty payments from Kite, a Gilead Company. A.B., J.R., A.X. and N.S. are all employees of Kite, a Gilead Company.
Additional information
Peer review information Saheli Sadanand was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 CONSORT.
Consort diagram of the Hu19-CD828Z clinical trial.
Extended Data Fig. 2 Hu19-CD828Z neurologic toxicities.
All Grade 4, 3, and 2 neurologic adverse events within the first month after CAR T-cell infusion are listed. Grading by National Cancer Institute Common Terminology Criteria for Adverse Events Version 3; all adverse events listed under “Neurologic” are included except syncope. Syncope was not included because it was associated with cytokine-release syndrome and hypotension. The highest grade of each adverse event experienced by each patient is listed. For example, if a patient had both Grade 2 and Grade 3 tremor at different times, tremor is only listed under Grade 3.
Extended Data Fig. 3 FMC63-28Z neurologic toxicities.
All Grade 4, 3, and 2 neurologic adverse events within the first month after CAR T-cell infusion are listed. Grading by National Cancer Institute Common Terminology Criteria for Adverse Events Version 3; all adverse events listed under “Neurologic” are included except syncope. Syncope was not included because it was associated with hypotension from cytokine-release syndrome. The highest grade of each adverse event experienced by each patient is listed. For example, if a patient had both Grade 2 and Grade 3 confusion at different times, confusion is only listed under Grade 3.
Extended Data Fig. 4 Peak serum protein levels.
For all proteins, all 22 patients on the trial of FMC63-28Z T cells and all 20 patients on the trial of Hu19-CD828Z T cells were compared. Proteins were measured in serum samples by Luminex® assay between day 2 and 14 after CAR T-cell infusion. Statistics were by 2-tailed Mann-Whitney test.
Extended Data Fig. 5 Patient 3 immunologic protein levels.
Patient 3 was the only patient with Grade 3 or 4 neurologic toxicity on the Hu19-CD828Z trial. Peak serum levels of 9 immunological proteins are shown for patient 3. Peak levels were determined between day 2 and day 14 after CAR T-cell infusion. These 9 proteins are shown because they were found to be prominently different between the Hu19-CD828Z and FMC63-28Z clinical trials (Fig. 3). Proteins were measured by Luminex® assay. MCP-1, monocyte chemotactic protein-1; IL, interleukin; TNF-alpha, tumor necrosis factor-alpha; MIP-1-alpha, macrophage inflammatory protein-1-alpha; IFN-gamma, interferon-gamma. The red bars indicate the median protein levels for all 20 patients that received Hu19-CD828Z CAR T cells.
Extended Data Fig. 6 Serum proteins areas under the curves.
For all proteins, all 22 patients on the trial of FMC63-28Z T cells and all 20 patients on the trial of Hu19-CD828Z T cells were compared. Proteins were measured in serum samples by Luminex® assay from days 2 to 14 after CAR T-cell infusion. Area under the curve (AUC) was calculated by trapezoidal method. Statistics were by 2-tailed Mann-Whitney test.
Extended Data Fig. 7 Structural Models of CARs.
Top row: schematic representations of Hu19-CD828Z (left) and FMC63-28Z (right) CAR models are shown; scFv in blue; hinge in green; transmembrane domain in yellow; intracellular domain in red. The membrane position during molecular dynamics simulations is shown in grey. Bottom row: conformational flexibility for each corresponding CAR depicted as superimposed carbon-alpha traces for a set of 50 representative conformations observed during a 50 nanosecond molecular dynamics trajectory. The differences in flexibility originate in the very different structure and dynamic behavior of the corresponding hinge regions during the dynamics simulations. Transmembrane and scFv domains are affected by the hinge properties and display very different behaviors as well. A quantitative analysis of the molecular dynamics trajectories reveals that these behaviors affect the scFv mobility (assessed as molecular diffusibility) and the proper formation of a transmembrane dimer evaluated by the helix-helix occluded surface. All models assume a dimeric structure anchored by disulfide bonds. In short, Hu19-CD828Z exhibited less conformational flexibility than FMC63-28Z.
Extended Data Fig. 8 Anti-CAR immune responses measured by ELISPOT.
*Positive anti-CAR response was defined as 3x or greater increase in spot number from pretreatment to post-CAR T-cell infusion, and post-treatment spot number must have been 3x or more than the spot number of the media control. #Bin A contained peptides from the signal sequences, scFv linker, and hinge regions. Bin B contained peptides from the scFv light chain. Bin C contained peptides from the scFv heavy chain. Bin D contained peptides from transmembrane and intracellular domains. ^The increase in spots was the number of spots/400,000 total input PBMC at the positive time-point minus the number of spots/400,000 total input PBMC before CAR T-cell treatment.
Extended Data Fig. 9 Blood CAR T-cell levels and anti-CAR immune responses.
Anti-CAR T-cell responses were assessed by ELISPOT analysis of PBMC before CAR T-cell treatment and at time-points within 6 weeks after CAR T-cell infusion as summarized in Extended Data Fig. 8. CAR+ cell levels in the blood were assessed by quantitative PCR. The top row shows peak blood CAR+ cell levels with results divided into patients with or without anti-CAR responses by ELISPOT. (a) Hu19-CD828Z (b) FMC63-28Z. The bottom row shows blood CAR+ cell levels 1-month after CAR T-cell infusion with results divided into patients with or without anti-CAR responses detected by ELISPOT: (c) Hu19-CD828Z, (d) FMC63-28Z. No statistically significant differences in blood CAR+ cell levels were found between patients with or without anti-CAR responses. All patients with adequate cell samples for both ELISPOT and qPCR are included. P values by Mann-Whitney test are shown on the plots; significance was defined as P<0.05. Of the 4 comparisons, the FMC63-28Z 1-month comparison was closest to statistical significance with P=0.061. Each symbol represents an individual patient. The number of unique patients analyzed were as follows: Hu19-CD828Z Peak, n=18; FMC63-28Z Peak, n=18; Hu19-CD828Z 1 month, n=18; FMC63-28Z 1 month n=13.
Supplementary information
Supplementary Information
Supplementary Figs. 1–7, Supplementary Tables 1–7 and Clinical Trial Protocols
Source data
Source Data Fig. 1
Statistical source data
Source Data Fig. 2
Statistical source data
Source Data Fig. 3
Statistical source data
Source Data Fig. 4
Statistical source data
Source Data Fig. 5
Statistical source data
Source Data Extended Data Fig. 5
Statistical source data
Source Data Extended Data Fig. 9
Statistical source data
Rights and permissions
About this article
Cite this article
Brudno, J.N., Lam, N., Vanasse, D. et al. Safety and feasibility of anti-CD19 CAR T cells with fully human binding domains in patients with B-cell lymphoma. Nat Med 26, 270–280 (2020). https://doi.org/10.1038/s41591-019-0737-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-019-0737-3
This article is cited by
-
Programmable synthetic receptors: the next-generation of cell and gene therapies
Signal Transduction and Targeted Therapy (2024)
-
Harnessing the potential of CAR-T cell therapy: progress, challenges, and future directions in hematological and solid tumor treatments
Journal of Translational Medicine (2023)
-
The screening, identification, design and clinical application of tumor-specific neoantigens for TCR-T cells
Molecular Cancer (2023)
-
Limited utility of chimeric antigen receptor (CAR) T-cell retreatment: experience with a human anti-CD19 CAR
Bone Marrow Transplantation (2023)
-
Long-term outcomes following CAR T cell therapy: what we know so far
Nature Reviews Clinical Oncology (2023)