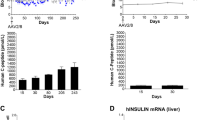
Abstract
In autoimmune (type 1) diabetes, autoreactive lymphocytes destroy pancreatic β-cells responsible for insulin synthesis. To assess the feasibility of gene therapy for type 1 diabetes, recombinant vaccinia virus (rVV) vectors were constructed expressing pancreatic islet autoantigens proinsulin (INS) and a 55-kDa immunogenic peptide from glutamic acid decarboxylase (GAD), and the immunomodulatory cytokine interleukin (IL)-10. To augment the beneficial effects of recombinant virus therapy, the INS and GAD genes were fused to the C terminus of the cholera toxin B subunit (CTB). Five-week-old non-obese diabetic (NOD) mice were injected once with rVV. Humoral antibody immune responses and hyperglycemia in the infected mice were analyzed.
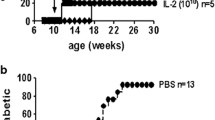
Only 20% of the mice inoculated with rVV expressing the CTB::INS fusion protein developed hyperglycemia, in comparison to 70% of the mice in the uninoculated animal group. Islets from pancreatic tissues isolated from euglycemic mice from this animal group showed no sign of inflammatory lymphocyte invasion. Inoculation with rVV producing CTB::GAD or IL-10 was somewhat less effective in reducing diabetes. Humoral antibody isotypes of hyperglycemic and euglycemic mice from all treated groups possessed similar IgG1/IgG2c antibody titer ratios from 19 to 32 wk after virus inoculation. In comparison with uninoculated mice, 11-wk-old NOD mice injected with virus expressing CTB::INS were delayed in diabetes onset by more than 4 wk. The experimental results demonstrate the feasibility of using rVV expressing CTB::INS fusion protein to generate significant protection and therapy against type 1 diabetes onset and progression.
Article PDF
Similar content being viewed by others
References
Eisenbarth, G. (1986) Type I diabetes mellitus: a chronic autoimmune disease. N. Eng. J. Med., 314, 1360–1368.
Tisch, R. and McDevitt, H. (1996) Insulin-dependent diabetes mellitus. Cell, 85, 291–297.
Czerkinsky, C., Anjuere, F., McGhee, J. R., et al. (1999) Mucosal immunity and tolerance: relevance to vaccine development. Immunol. Rev., 170, 197–222.
Bregenholt, S., Wang, M., Wolfe, T., et al. (2003) The choleratoxin B subunit is a mucosal adjuvant for oral tolerance induction in type 1 diabetes. Scand. J. Immunol, 57, 432–438.
Holmgren, J., Lycke, N., and Czerkinsky, C. (1993). Choleratoxin and cholera B subunit asoral-mucosal adjuvant and antigen vector systems. Vaccine 11, 1179–1184.
Sadeghi, H., Bregenholt, S., Wegmannn, D., et al. (2002) Genetic fusion of human insulin B-chain to the B-subunit of choleratoxin enhances in vitro antigen presentation and induction of bystander suppression in vivo. Immunology, 106, 237–245.
Sun, J. B., Holmgren, J., and Czerkinsky, C. (1994) Cholera toxin B subunit an efficient transmucosal carrier-delivery system for induction of peripheral immunological tolerance Proc. Natl. Acad. Sci. USA 91, 10,795–10,799.
Nagamatsu, S., Nakamichi, Y., Ohara-Imaizumi, M., et al. (2001) Adenovirus-mediated preproinsulin gene transfer into adipose tissues ameliorates hyperglycemia in obese diabetic KKA(y) mice FEBS Lett. 59, 106–110.
Jun, H. S., Khil, L. Y., and Yoon, J. W. (2002) Role of glutamic acid decarboxylase in the pathogenesis of type 1 diabetes. Cell. Mol. Life Sci 59, 1892–1901.
Hang, G., Li, Y., Wang, J., et al. (2005) Active tolerance induction and prevention of autoimmune diabetes by immunogene therapy using recombinant adenoassociated virus expressing glutamic acid decarboxylase 65 peptide GAD (500–585). J. Immnol. 174, 4516–4524.
Goudy, K., Song, S., Wasserfall, C., et al. (2001) Adeno-associated virus vector-mediated IL-10 gene delivery prevents type 1 diabetes in NOD mice. Proc. Natl. Acad. Sci. USA 98, 13,913–13,918.
Yang, Z., Chen, M., Fialkow, L. B., et al. (2000) Suppression of autoimmune diabetes by viral IL-10 gene transfer. J. Immunol. 168, 6479–6485.
Zhang, Y. C., Pileggi, A., Agarwal, A., et al. (2003) Adeno-associated virus-mediated IL-10 gene therapy inhibitsdiabetes recurrence in syngeneic islet cell transplantation of NOD mice. Diabetes 52, 708–716.
Oehen, S., Ohashi, P. S., Aichele, P., et al. (1992) Vaccination or tolerance to prevent diabetes. Eur. J. Immunol. 22, 3149–3153.
Ohashi, P. S., Oehen, S., Aichele, P., et al. (1993) Induction of diabetes in influenced by the infectious virus and local expression of MHC class I and tumor necrosis factor-alpha. J. Immunol. 150, 5185–5194.
Jun, H. S., Chung, Y. H., Han, J., et al. (2002) Prevention of autoimmune diabetes by immunogene therapy using recombinant vaccinia virus expressing glutamic acid decarboxylase. Diabetologia 45, 668–676.
Denes, B., Krausova, V., Yu J., et al. (2005) Recombinant vaccinia viruses containing cholera toxin-islet autoantigen fusion genes protect NOD mice against autoimmune diabetes. J. Immunother. 28, 438–448.
Timiryasova, T. M., Chen, B., Haghighat, P., et al. (1999) Vaccinia virus-mediated expression of wild-type p53 suppresses glioma cell growth and induces apoptosis. Oncol. Res. 11, 133–144.
Chakrabarti, S., Sisler, J. R., and Moss, B. (1997) Compact, synthetic, vaccinia virus early/late promoter for protein expression. BioTechniques 23, 1094–1097.
Timiryasova, T. M., Chen, B., and Fodor, I. (2001) Replication-deficient vaccinia virus gene therapy vector: evaluation of exogenous gene expression mediated by PUV-inactivated virus in glioma cells. J. Gene Med. 3, 468–477.
Zhang, J., Davidson, L., Eisenbarth, G., et al. (1991) Suppression of diabetes in nonobese diabetic mice by oral administration of porcine insulin. Proc. Natl. Acad. Sci. USA 88, 10,252–10,256.
Arakawa, T., Chong, D. K., Merritt, J. L., et al. (1997) Expression of cholera toxin B subunit oli gomers in transgenic potato plants. Transgenic Res. 6, 403–413.
Carter, J. D., Ellett, J. D., Chen, M., et al. (2005) Virall IL-10-mediated immune regulation in pancreatic islet transplantation. Mol. Ther. 12, 360–368.
Nakayama, M., Abiru, N., Moriyama, H., et al. (2005) Prime role for an insulin epitope in the development of type 1 diabetes in NOD mice. Nature 435, 220–223.
Kent, S. C., Chen, Y., Bregoli, L., et al. 2005 Expanded T cells from pancreatic lymphnodes of type 1 diabetic subjects recognize an insulin epitope. Nature 435, 224–228.
Yang, Z., Chen, M., Wu, R., et al. (2002) Suppression of autoimmune diabetes by viral IL-10 gene transfer. J. Immunol 168, 6479–6485.
Moss, B. (1991) Vaccinia virus: a tool for research and development. Science 252, 1660–1667.
Moss, B. (1996) Genetically engineered poxvirus for recombinant gene expression, vaccination and safety, Proc. Natl. Acad. Sci. USA 93, 11,341–11,348.
Taylor, G., Stott, E. J., Wertz, G., et al. (1991) Comparison of the virulence of wild-type thymidine kinase (tk)-deficient and tk+phenotypes of vaccinia virus recombinants after intranasal inoculation of mice. J. Gen. Virol. 72, 125–130.
Verardi, P. H., Jones, L. A., Aziz, F. H., et al. (2001) Vaccinia virus vectors with an inactivated gamma interferon receptor homolog gene (B8R) are attenuated in vivo without a concomitant reduction in immunogenicity. J. Virol. 75, 11–18.
Bender, B. S., Rowe, C. A., Taylor, S. F., et al. (1996) Oral immunization with a replication-deficient recombinant vaccinia virus protects mice against influenza. J. Virol. 70, 6418–6424.
Etchart, N., Wild, F., and Kaiserlian, D. (1996) Mucosal and systemic immune responses to measles virus haemagglutinin in mice immunized with a recombinant vaccinia virus. J. Gen. Virol. 10, 2471–2478.
Arakawa, T., Chong, D. K. X., Yu, J., et al. (1998) A plant-based cholera toxin B subunit-insulin fusion protein protects against development of autoimmune daiabetes. Nat. Biotechnol. 16, 934–938.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dénes, B., Yu, J., Fodor, N. et al. Suppression of hyperglycemia in NOD mice after inoculation with recombinant vaccinia viruses. Mol Biotechnol 34, 317–327 (2006). https://doi.org/10.1385/MB:34:3:317
Issue Date:
DOI: https://doi.org/10.1385/MB:34:3:317